Medroxyprogesterone (MP), is a progestin which is not used medically. A derivative, medroxyprogesterone acetate (MPA), is used as a medication in humans, and is far more widely known in comparison. Medroxyprogesterone is sometimes used as a synonym for medroxyprogesterone acetate, and what is almost always being referred to when the term is used is MPA and not medroxyprogesterone.

Canrenone, sold under the brand names Contaren, Luvion, Phanurane, and Spiroletan, is a steroidal antimineralocorticoid of the spirolactone group related to spironolactone which is used as a diuretic in Europe, including in Italy and Belgium. It is also an important active metabolite of spironolactone, and partially accounts for its therapeutic effects.

Cyproterone, also known by its developmental code name SH-80881, is a steroidal antiandrogen which was studied in the 1960s and 1970s but was never introduced for medical use. It is an analogue of cyproterone acetate (CPA), an antiandrogen, progestin, and antigonadotropin which was introduced instead of cyproterone and is widely used as a medication. Cyproterone and CPA were among the first antiandrogens to be developed.

Delmadinone acetate (DMA), sold under the brand name Tardak among others, is a progestin and antiandrogen which is used in veterinary medicine to treat androgen-dependent conditions such as benign prostatic hyperplasia. It must be used with care as it has the potential to cause adrenal insufficiency via inhibition of adrenocorticotropic hormone (ACTH) secretion from the pituitary gland. DMA is the C17α acetate ester of delmadinone, which, in contrast to DMA, was never marketed for medical use.

Benorterone, also known by its developmental code name SKF-7690 and as 17α-methyl-B-nortestosterone, is a steroidal antiandrogen which was studied for potential medical use but was never marketed. It was the first known antiandrogen to be studied in humans. It is taken by mouth or by application to skin.
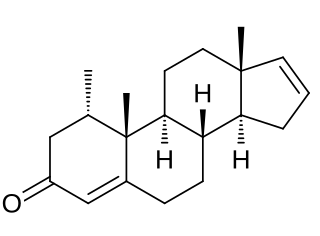
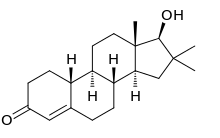
Delanterone (INN), also known as 1α-methylandrosta-4,16-dien-3-one, is a steroidal antiandrogen described as an anti-acne agent which was never marketed. The compound showed poor efficacy as an antiandrogen in vivo in animals, suggestive of low activity or a short terminal half-life, and likely in relation to this was not further developed. It was described and characterized in the literature in 1977.
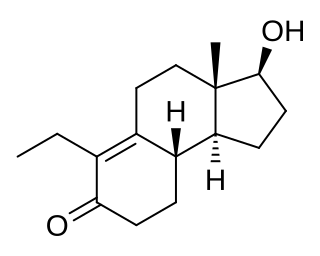
Inocoterone is a steroid-like nonsteroidal antiandrogen (NSAA) that was never marketed. An acetate ester, inocoterone acetate, shows greater antiandrogen activity and was developed as a topical medication for the treatment of acne but showed only modest effectiveness in clinical trials and similarly was never marketed.
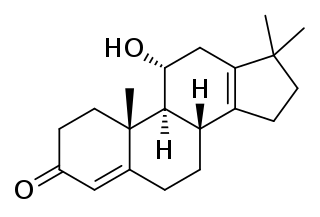
Nordinone (INN), also known as 11α-hydroxy-17,17-dimethyl-18-norandrosta-4,13-dien-3-one, is a naturally occurring steroid with antiandrogen properties isolated as a metabolite from the fungus Monocillium nordinii.

Oxendolone, sold under the brand names Prostetin and Roxenone, is an antiandrogen and progestin medication which is used in Japan in the treatment of enlarged prostate. However, this use is controversial due to concerns about its clinical efficacy. Oxendolone is not effective by mouth and must be given by injection into muscle.

Topterone, also known as 17α-propyltestosterone or as 17α-propylandrost-4-en-17β-ol-3-one, is a steroidal antiandrogen that was first reported in 1978 and was developed for topical administration but, due to poor effectiveness, was never marketed.
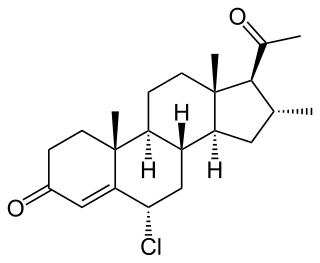
Clometerone (INN), or clometherone (USAN), also known as 6α-chloro-16α-methylprogesterone, is a synthetic pregnane steroid and derivative of progesterone which was reported in 1962 and is described as an antiestrogen and antiandrogen but was never marketed.

Mespirenone (INN), also known as Δ1-15β,16β-methylenespironolactone, is a steroidal antimineralocorticoid of the spirolactone group related to spironolactone that was never marketed. Animal research found that it was 3.3-fold more potent as an antimineralocorticoid relative to spironolactone. In addition to its antimineralocorticoid properties, mespirenone is also a progestogen, antigonadotropin, and antiandrogen. It is 2- to 3-fold as potent as spironolactone as a progestogen and antigonadotropin but its antiandrogenic activity is markedly reduced and weak in comparison. Mespirenone is also a potent and specific enzyme inhibitor of 18-hydroxylase and thus of mineralocorticoid biosynthesis. The drug was under development by Schering and reached phase II clinical trials but was discontinued in 1989.

Spirorenone (INN) is a steroidal antimineralocorticoid of the spirolactone group that was never marketed. Spirorenone possesses 5–8 times the antimineralocorticoid activity of spironolactone in animal studies. The initial discovery of spirorenone was deemed a great success, as no compound with greater antimineralocorticoid activity had been developed since spironolactone in 1957. Moreover, spirorenone itself has virtually no affinity for the androgen receptor while its progestogenic activity shows species differences, being somewhat greater than that of spironolactone in rabbits but absent in mice and rats. As such, it was characterized as a highly potent antimineralocorticoid with far fewer hormonal side effects relative to spironolactone.

Bifluranol is a synthetic nonsteroidal estrogen of the stilbestrol group related to diethylstilbestrol that has been used as an antiandrogen in the United Kingdom in the treatment of benign prostatic hyperplasia. It is a polyfluorinated biphenyl that is related to polybrominated and polychlorinated biphenyls and diethylstilbestrol. The drug is described as a weak estrogen, and possesses about one-eighth the potency of diethylstilbestrol.

Edogestrone, or edogesterone, also known as 17α-acetoxy-3,3-ethylenedioxy-6-methylpregn-5-en-20-one, is a steroidal progestin and antiandrogen of the 17α-hydroxyprogesterone group which was synthesized in 1964 but was never marketed. Similarly to the structurally related steroid cyproterone acetate, edogestrone binds directly to the androgen receptor and antagonizes it, displacing androgens like testosterone from the receptor, though not as potently as cyproterone acetate. The drug has also been found to suppress androgen production, likely via progesterone receptor activation-mediated antigonadotropic activity.

Amadinone (INN), also known as 19-norchlormadinone, is a steroidal progestin of the 19-norprogesterone and 17α-hydroxyprogesterone groups that was synthesized and characterized in 1968 but was never marketed. It has antigonadotropic properties, and for this reason, is a functional antiandrogen. An acetate ester, amadinone acetate, also exists, but similarly was never marketed.

BOMT, also known by its developmental code name Ro 7-2340 and as 6α-bromo-4-oxa-17α-methyl-5α-dihydrotestosterone, is a synthetic steroidal antiandrogen which was first developed in 1970 and was never marketed for medical use. It is the 6α-brominated, 4-oxygenated, and 17α-methylated derivative of the androgen dihydrotestosterone (DHT). Along with benorterone, cyproterone, and flutamide, BOMT was among the earliest antiandrogens to be developed and extensively studied, although it is less well-documented in comparison to the others. BOMT has been investigated clinically in the treatment of benign prostatic hyperplasia, though development for this use did not continue. There was also interest in BOMT for the potential applications of acne, pattern hair loss, and possibly prostate cancer, but it was not developed for these indications either.
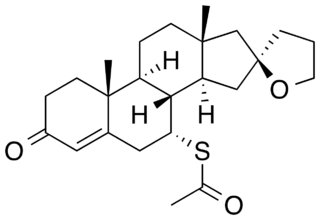
Spiroxasone is a synthetic, steroidal antimineralocorticoid of the spirolactone group which was developed as a diuretic and antihypertensive agent but was never marketed. It was synthesized and assayed in 1963. The drug is 7α-acetylthiospirolactone with the ketone group removed from the C17α spirolactone ring. Similarly to other spirolactones like spironolactone, spiroxasone also possesses antiandrogen activity.

Dicirenone is a synthetic, steroidal antimineralocorticoid of the spirolactone group which was developed as a diuretic and antihypertensive agent but was never marketed. It was synthesized and assayed in 1974. Similarly to other spirolactones like spironolactone, dicirenone also possesses antiandrogen activity, albeit with relatively reduced affinity.

7α-Thioprogesterone is a synthetic, steroidal, and potent antimineralocorticoid (putative) and antiandrogen which was developed by G. D. Searle & Co and was described in the late 1970s and early 1980s but was never developed or introduced for medical use. It is a derivative of progesterone (pregn-4-ene-3,20-dione) with a thio (sulfur) substitution at the C7α position, and is related to the spirolactone group of drugs but lacks a γ-lactone ring.