| |
| Clinical data | |
|---|---|
| Other names | Ethynodiol; 3β-Hydroxynorethisterone; 17α-Ethynylestr-4-ene-3β,17β-diol |
| Drug class | Progestin; Progestogen |
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.013.610 |
| Chemical and physical data | |
| Formula | C20H28O2 |
| Molar mass | 300.442 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
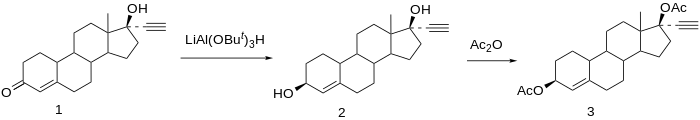
Etynodiol, or ethynodiol, is a steroidal progestin of the 19-nortestosterone group which was never marketed. [1] [2] [3] A diacylated derivative, etynodiol diacetate, is used as a hormonal contraceptive. [1] [2] Etynodiol is sometimes used as a synonym for etynodiol diacetate.
Contents
It was patented in 1955. [4]