Selective androgen receptor modulators (SARMs) are a class of drugs that selectively activate the androgen receptor in specific tissues, promoting muscle and bone growth while having less effect on male reproductive tissues like the prostate gland.

BMS-564,929 is an investigational selective androgen receptor modulator (SARM) which is being developed by Bristol-Myers Squibb for treatment of the symptoms of age-related decline in androgen levels in men ("andropause"). These symptoms may include depression, loss of muscle mass and strength, reduction in libido and osteoporosis. Treatment with exogenous testosterone is effective in counteracting these symptoms but is associated with a range of side effects, the most serious of which is enlargement of the prostate gland, which can lead to benign prostatic hypertrophy and even prostate cancer. This means there is a clinical need for selective androgen receptor modulators, which produce anabolic effects in some tissues such as muscle and bone, but without stimulating androgen receptors in the prostate.

S-40503 is an investigational selective androgen receptor modulator (SARM) developed by the Japanese company Kaken Pharmaceuticals, which was developed for the treatment of osteoporosis. SARMs are a new class of drugs which produce tissue-specific anabolic effects in some tissues such as muscle and bone, but without stimulating androgen receptors in other tissues such as in the prostate gland, thus avoiding side effects such as benign prostatic hypertrophy which can occur following treatment with unselective androgens like testosterone or anabolic steroids.

Enobosarm, also formerly known as ostarine and by the developmental code names GTx-024, MK-2866, and S-22, is a selective androgen receptor modulator (SARM) which is under development for the treatment of androgen receptor-positive breast cancer in women and for improvement of body composition in people taking GLP-1 receptor agonists like semaglutide. It was also under development for a variety of other indications, including treatment of cachexia, Duchenne muscular dystrophy, muscle atrophy or sarcopenia, and stress urinary incontinence, but development for all other uses has been discontinued. Enobosarm was evaluated for the treatment of muscle wasting related to cancer in late-stage clinical trials, and the drug improved lean body mass in these trials, but it was not effective in improving muscle strength. As a result, enobosarm was not approved and development for this use was terminated. Enobosarm is taken by mouth.
LGD-3303 is a drug which acts as a selective androgen receptor modulator (SARM), with good oral bioavailability. It is a selective agonist for the androgen receptor, producing functional selectivity with effective dissociation of anabolic and androgenic effects, acting as a partial agonist for androgenic effects, but a full agonist for anabolic effects. It has been investigated as a possible treatment for osteoporosis, and was shown in animal studies to enhance the effectiveness of a bisphosphonate drug.
GTx, Inc. was a pharmaceutical company that is working on drugs in the selective estrogen receptor modulator (SERM) and selective androgen receptor modulator (SARM) classes. Its drugs in development included enobosarm (ostarine) and GTx-758.
The first antiandrogen was discovered in the 1960s. Antiandrogens antagonise the androgen receptor (AR) and thereby block the biological effects of testosterone and dihydrotestosterone (DHT). Antiandrogens are important for men with hormonally responsive diseases like prostate cancer, benign prostatic hyperplasia (BHP), acne, seborrhea, hirsutism and androgen alopecia. Antiandrogens are mainly used for the treatment of prostate diseases. Research from 2010 suggests that ARs could be linked to the disease progression of triple-negative breast cancer and salivary duct carcinoma and that antiandrogens can potentially be used to treat it.

LGD-4033, also known by the developmental code name VK5211 and by the black-market name Ligandrol, is a selective androgen receptor modulator (SARM) which is under development for the treatment of muscle atrophy in people with hip fracture. It was also under development for the treatment of cachexia, hypogonadism, and osteoporosis, but development for these indications was discontinued. LGD-4033 has been reported to dose-dependently improve lean body mass and muscle strength in preliminary clinical trials, but is still being developed and has not been approved for medical use. The drug is taken by mouth.

A nonsteroidal antiandrogen (NSAA) is an antiandrogen with a nonsteroidal chemical structure. They are typically selective and full or silent antagonists of the androgen receptor (AR) and act by directly blocking the effects of androgens like testosterone and dihydrotestosterone (DHT). NSAAs are used in the treatment of androgen-dependent conditions in men and women. They are the converse of steroidal antiandrogens (SAAs), which are antiandrogens that are steroids and are structurally related to testosterone.

Vosilasarm, also known by the development codes RAD140 and EP0062 and by the black-market name Testolone or Testalone, is a selective androgen receptor modulator (SARM) which is under development for the treatment of hormone-sensitive breast cancer. It is specifically under development for the treatment of androgen receptor-positive, estrogen receptor-negative, HER2-negative advanced breast cancer. Vosilasarm was also previously under development for the treatment of sarcopenia, osteoporosis, and weight loss due to cancer cachexia, but development for these indications was discontinued. The drug is taken by mouth.

Acetothiolutamide is a selective androgen receptor modulator (SARM) derived from the nonsteroidal antiandrogen bicalutamide that was described in 2002 and was one of the first SARMs to be discovered and developed. It is a high-affinity, selective ligand of the androgen receptor (AR), where it acts as a full agonist in vitro, and has in vitro potency comparable to that of testosterone. However, in vivo, acetothiolutamide displayed overall negligible androgenic effects, though significant anabolic effects were observed at high doses. In addition, notable antiandrogen effects were observed in castrated male rats treated with testosterone propionate. The discrepancy between the in vitro and in vivo actions of acetothiolutamide was determined to be related to rapid plasma clearance and extensive hepatic metabolism into a variety of metabolites with differing pharmacological activity, including AR partial agonism and antagonism. In accordance with its poor metabolic stability, acetothiolutamide is not orally bioavailable, and shows activity only via injected routes such as subcutaneous and intravenous.

LG121071 is a selective androgen receptor modulator (SARM) developed by Ligand Pharmaceuticals that was first described in 1999 and was the first orally active nonsteroidal androgen to be discovered. It is a tricyclic quinolone derivative, structurally distinct from other nonsteroidal AR agonists like andarine and enobosarm (ostarine). The drug acts as a high-affinity full agonist of the androgen receptor (AR), with a potency and efficacy that is said to be equivalent to that of dihydrotestosterone (DHT). Unlike testosterone, but similarly to DHT, LG121071 and other nonsteroidal androgens cannot be potentiated by 5α-reductase in androgenic tissues, and for this reason, show tissue-selective androgenic effects. In accordance, they are said to possess full anabolic activity with reduced androgenic activity, similarly to anabolic-androgenic steroids.

GTx-758 is a synthetic nonsteroidal estrogen which was under development by GTx, Inc. for the treatment of advanced prostate cancer. As of 2016, it had completed two phase II clinical trials.

LG-120907 is a nonsteroidal antiandrogen (NSAA) of the quinoline group which was developed by Ligand Pharmaceuticals along with selective androgen receptor modulators (SARMs) like LG-121071 and was never marketed. The drug is a high-affinity antagonist of the androgen receptor (AR) with a Ki value of 26 nM and has been found to inhibit growth of the ventral prostate and seminal vesicles in male rats without increasing circulating levels of luteinizing hormone or testosterone. However, this tissue selectivity has not been assessed in humans. LG-120907 is orally active and shows greater oral potency than the arylpropionamide NSAA flutamide.

The pharmacology of bicalutamide is the study of the pharmacodynamic and pharmacokinetic properties of the nonsteroidal antiandrogen (NSAA) bicalutamide. In terms of pharmacodynamics, bicalutamide acts as a selective antagonist of the androgen receptor (AR), the biological target of androgens like testosterone and dihydrotestosterone (DHT). It has no capacity to activate the AR. It does not decrease androgen levels and has no other important hormonal activity. The medication has progonadotropic effects due to its AR antagonist activity and can increase androgen, estrogen, and neurosteroid production and levels. This results in a variety of differences of bicalutamide monotherapy compared to surgical and medical castration, such as indirect estrogenic effects and associated benefits like preservation of sexual function and drawbacks like gynecomastia. Bicalutamide can paradoxically stimulate late-stage prostate cancer due to accumulated mutations in the cancer. When used as a monotherapy, bicalutamide can induce breast development in males due to its estrogenic effects. Unlike other kinds of antiandrogens, it may have less adverse effect on the testes and fertility.

RU-59063 is a nonsteroidal androgen or selective androgen receptor modulator (SARM) which was first described in 1994 and was never marketed. It was originally thought to be a potent antiandrogen, but subsequent research found that it actually possesses dose-dependent androgenic activity, albeit with lower efficacy than dihydrotestosterone (DHT). The drug is an N-substituted arylthiohydantoin and was derived from the first-generation nonsteroidal antiandrogen (NSAA) nilutamide. The second-generation NSAAs enzalutamide, RD-162, and apalutamide were derived from RU-59063.

β-LGND2, also known as ERβ-selective ligand 2 or as GTx-878, is a synthetic nonsteroidal estrogen and selective ERβ agonist which was under development by GTx for the treatment of benign prostatic hyperplasia, prostatitis, and rheumatoid arthritis but was never marketed. It shows approximately 25-fold selectivity for activation of the ERβ over the ERα (EC50Tooltip half-maximal effective concentration = 2 nM and 52 nM, respectively). β-LGND2 is an isoquinolinone derivative.

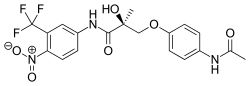
Acetoxolutamide is a nonsteroidal androgen and selective androgen receptor modulator (SARM) which was described in 2000 and was never developed or marketed for medical use. It was derived from structural modification of the nonsteroidal antiandrogen bicalutamide and the nonsteroidal SARM acetothiolutamide. Acetoxolutamide shows greatly improved pharmacokinetic properties and anabolic and androgenic potency relative to acetothiolutamide in animals. It is the (2R) enantiomer of andarine.

JNJ-37654032 is a selective androgen receptor modulator (SARM) which was developed by Johnson & Johnson for the potential treatment of muscular atrophy but was never marketed.

GTx-027 is a selective androgen receptor modulator (SARM) which was under development for or of potential interest in the treatment of breast cancer and stress urinary incontinence (SUI) but was never marketed. It is taken by mouth.