 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Noristerat, others |
| Other names | NETE; NET-EN; Norethindrone enanthate; SH-393; 17α-Ethynyl-19-nortestosterone 17β-enanthate; 17α-Ethynylestra-4-en-17β-ol-3-one 17β-enanthate |
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | Intramuscular injection |
| Drug class | Progestogen; Progestin; Progestogen ester |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.021.207 |
| Chemical and physical data | |
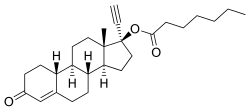
| Formula | C27H38O3 |
| Molar mass | 410.598 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Norethisterone enanthate (NETE), also known as norethindrone enanthate, is a form of hormonal birth control which is used to prevent pregnancy in women. [1] [2] [3] It is used both as a form of progestogen-only injectable birth control and in combined injectable birth control formulations. It may be used following childbirth, miscarriage, or abortion. [1] The failure rate per year in preventing pregnancy for the progestogen-only formulation is 2 per 100 women. [4] Each dose of this form lasts two months with only up to two doses typically recommended. [5] [1]
Contents
- Medical uses
- Contraindications
- Side effects
- Overdose
- Interactions
- Pharmacology
- Pharmacodynamics
- Pharmacokinetics
- Chemistry
- History
- Society and culture
- Generic names
- Brand names
- Availability
- Research
- See also
- References
- External links
Side effects include breast pain, headaches, depression, irregular menstrual periods, and pain at the site of injection. [5] Use in those with liver disease is not recommended as is use during pregnancy due to risk of birth defects. [1] Use appears to be okay during breastfeeding. [1] It does not protect against sexually transmitted infections. [1] NETE is an ester and prodrug of norethisterone, [6] through which it works. [1] It works as a method of birth control by stopping ovulation. [1]
Norethisterone was patented in 1951 and NETE came into medical use in 1957. [7] [8] It is on the World Health Organization's List of Essential Medicines. [9] It has been approved by itself in more than 60 countries including the United Kingdom and some in Europe, Central America, and Africa, and in combination with estradiol valerate in at least 36 countries mainly in Latin America. [4] [10] [11] [12] It is not available in the United States. [10]


