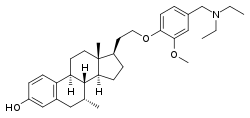
 | |
| Clinical data | |
|---|---|
| Other names | 17β-[2-[4-[(diethylamino)methyl]-2-methoxyphenoxy]ethyl]-7α-methylestra-1,3,5(10)-trien-3-ol; 17β-[2-[4-[(diethylamino)methyl]-2-methoxyphenoxy]ethyl]-7α-methylestradiol |
| Routes of administration | By mouth [1] |
| ATC code | |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| UNII |
|
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C33H47NO3 |
| Molar mass | 505.743 g·mol−1 |
| 3D model (JSmol) |
|
| |
| |
TAS-108, also known as SR-16234, is a drug discovered by Masato Tanabe and under development by SRI International and Taiho Pharmaceutical. It is a steroid hormone that has shown signs of treating and preventing breast cancer, even in patients where tamoxifen has failed. [2] [3]