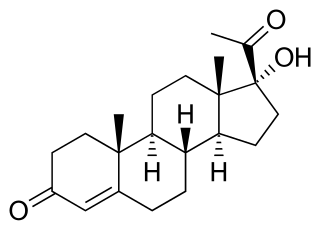
17α-Hydroxyprogesterone (17α-OHP), also known as 17-OH progesterone (17-OHP), or hydroxyprogesterone (OHP), is an endogenous progestogen steroid hormone related to progesterone. It is also a chemical intermediate in the biosynthesis of many other endogenous steroids, including androgens, estrogens, glucocorticoids, and mineralocorticoids, as well as neurosteroids.

Medroxyprogesterone (MP), is a progestin which is not used medically. A derivative, medroxyprogesterone acetate (MPA), is used as a medication in humans, and is far more widely known in comparison. Medroxyprogesterone is sometimes used as a synonym for medroxyprogesterone acetate, and what is almost always being referred to when the term is used is MPA and not medroxyprogesterone.
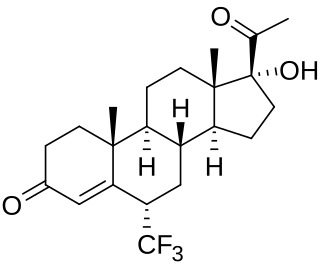
Flumedroxone is a steroidal progestogen of the 17α-hydroxyprogesterone group that was never marketed. The C17α acetate ester of flumedroxone, flumedroxone acetate, has been marketed as an antimigraine drug.

Melengestrol acetate (MLGA), sold under the brand names Heifermax and MGA among others, is a progestin medication which is used in animal reproduction. It is not approved for use in humans, and is instead used as an implantable contraceptive for captive animals in zoos and other refuges, and is also used as a feed additive to promote growth in cattle, a purpose it is licensed for in the United States and Canada.

Delmadinone acetate (DMA), sold under the brand name Tardak among others, is a progestin and antiandrogen which is used in veterinary medicine to treat androgen-dependent conditions such as benign prostatic hyperplasia. It must be used with care as it has the potential to cause adrenal insufficiency via inhibition of adrenocorticotropic hormone (ACTH) secretion from the pituitary gland. DMA is the C17α acetate ester of delmadinone, which, in contrast to DMA, was never marketed for medical use.

Hydroxyprogesterone acetate (OHPA), sold under the brand name Prodox, is an orally active progestin related to hydroxyprogesterone caproate (OHPC) which has been used in clinical and veterinary medicine. It has reportedly also been used in birth control pills.
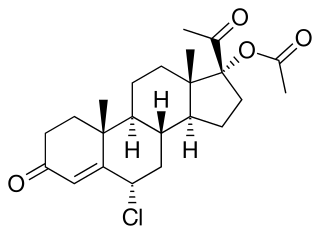
Hydromadinone acetate, also known as chloroacetoxyprogesterone (CAP), as well as 6α-chloro-17α-acetoxyprogesterone or 6α-chloro-17α-acetoxypregn-4-ene-3,20-dione, is a steroidal progestin of the 17α-hydroxyprogesterone group that was never marketed. It is the C17α acetate ester of hydromadinone, which, similarly, was never marketed.

Hydroxyprogesterone heptanoate (OHPH), also known as hydroxyprogesterone enanthate (OHPE) and sold under the brand names H.O.P., Lutogil A.P., and Lutogyl A.P. among others, is a progestin medication used for progestogenic indications. It has been formulated both alone and in together with estrogens, androgens/anabolic steroids, and other progestogens in several combination preparations. OHPH is given by injection into muscle at regular intervals.

Anagestone acetate, sold under the brand names Anatropin and Neo-Novum, is a progestin medication which was withdrawn from medical use due to carcinogenicity observed in animal studies.

A progestogen ester is an ester of a progestogen or progestin. The prototypical progestogen is progesterone, an endogenous sex hormone. Esterification is frequently employed to improve the pharmacokinetics of steroids, including oral bioavailability, lipophilicity, and elimination half-life. In addition, with intramuscular injection, steroid esters are often absorbed more slowly into the body, allowing for less frequent administration. Many steroid esters function as prodrugs.

Cismadinone acetate, also known as 6α-chloro-δ1-dehydro-17α-acetoxyprogesterone or as 6α-chloro-17α-acetoxypregna-1,4-diene-3,20-dione, is a steroidal progestin related to the 17α-hydroxyprogesterone derivatives which was never marketed. It is the acetylated form of cismadinone, which is also a progestin but, similarly to cismadinone acetate, was never marketed.

17α-Methylprogesterone (17α-MP), or 17α-methylpregn-4-ene-3,20-dione, is a steroidal progestin related to progesterone that was synthesized and characterized in 1949 but was never marketed. Along with ethisterone (1938) and 19-norprogesterone (1951), 17α-MP was one of the earliest derivatives of progesterone to be identified as possessing progestogenic activity. Similarly to progesterone and derivatives like 17α-hydroxyprogesterone and 19-norprogesterone, 17α-MP was found to possess poor oral bioavailability, but showed improved progestogenic activity relative to progesterone when administered via other routes. In addition to its activity as a progestogen, 17α-MP has also been found to possess some antiglucocorticoid activity.
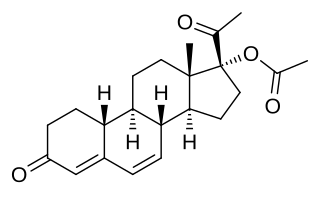
Gestadienol acetate an orally active progestin which was described in the literature in 1967 and was never marketed. It has no androgenic or estrogenic effects. The effects of gestadienol acetate on the endometrium and its general pharmacology were studied in a clinical trial in women. It has also been studied in a clinical trial for benign prostatic hyperplasia in men, but was ineffective.

Megestrol caproate, abbreviated as MGC, is a progestin medication which was never marketed. It was developed in Russia in 2002. In animals, MGC shows 10-fold higher progestogenic activity compared to progesterone when both are administered via subcutaneous injection. In addition, MGC has no androgenic, anabolic, or estrogenic activity. The medication was suggested as a potential contraceptive and therapeutic agent.

Methenmadinone acetate (MMA), also known as methylenedehydroacetoxyprogesterone (MDAP) and sold under the brand names Superlutin and Antigest, is a progestin medication which was developed in Czechoslovakia in the 1960s. It is the C17α acetate ester of methenmadinone.

16-Methylene-17α-hydroxyprogesterone acetate is a progestin of the 17α-hydroxyprogesterone group which was never marketed. Given orally, it shows about 2.5-fold the progestogenic activity of parenteral progesterone in animal bioassays. It is a parent compound of the following clinically used progestins:

6α-Methylprogesterone (6α-MP) is a progestin which was never marketed. It has 150% of the progestogenic potency of progesterone. In addition, and in contrast to progesterone, 6α-MP has weak androgenic, antiandrogenic, and synandrogenic actions. 6α-MP is structurally related to medroxyprogesterone acetate and megestrol acetate, which possess androgenic and/or antiandrogenic activity to varying degrees similarly. MPA is more androgenic than 6α-MP and MGA.

















