 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Andractim, others |
| Other names | Stanolone; Dihydrotestosterone; DHT; 5α-Dihydrotestosterone; 5α-DHT |
| Pregnancy category |
|
| Routes of administration | Transdermal (gel), in the cheek, under the tongue, intramuscular injection (as esters) |
| Drug class | Androgen; Anabolic steroid |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | Oral: Very low [2] Transdermal: 10% [2] [3] IM injection: 100% [3] |
| Metabolism | Liver |
| Elimination half-life | Transdermal: 2.8 hours [4] |
| Excretion | Urine |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
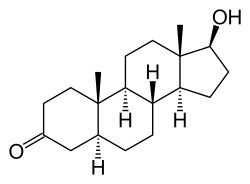
| Formula | C19H30O2 |
| Molar mass | 290.447 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Androstanolone, or stanolone, also known as dihydrotestosterone (DHT) and sold under the brand name Andractim among others, is an androgen and anabolic steroid (AAS) medication and hormone which is used mainly in the treatment of low testosterone levels in men. [2] It is also used to treat breast development and small penis in males. [2] Compared to testosterone, androstanolone (DHT) is less likely to aromatize into estrogen, and therefore it shows less pronounced estrogenic side effects, such as gynecomastia and water retention. On the other hand, androstanolone (DHT) show more significant androgenic side effects, such as acne, hair loss and prostate enlargement.
Contents
- Medical uses
- Available forms
- Side effects
- Pharmacology
- Pharmacodynamics
- Pharmacokinetics
- Chemistry
- Esters
- Derivatives
- History
- Society and culture
- Generic names
- Brand names
- Availability
- Legal status
- Research
- References
- External links
It has strong androgenic effects and muscle-building effects, as well as relatively weak estrogenic effects. [2]
It is typically given as a gel for application to the skin, but can also be used as an ester by injection into muscle. [2] [5]
Side effects of androstanolone include symptoms of masculinization like acne, increased hair growth, voice changes, and increased sexual desire. [2] The medication is a naturally occurring androgen and anabolic steroid and hence is an agonist of the androgen receptor (AR), the biological target of androgens like testosterone and DHT. [2] [6]
Androstanolone was discovered in 1935 and was introduced for medical use in 1953. [2] [7] [8] [9] It is used mostly in France and Belgium. [2] [10] [11] The drug has been used by weightlifters to increase performance due to its powerful androgenic properties. [12] [13] The medication is a controlled substance in many countries and so non-medical use is generally not permitted. [2]