Anticonvulsants are a diverse group of pharmacological agents used in the treatment of epileptic seizures. Anticonvulsants are also increasingly being used in the treatment of bipolar disorder and borderline personality disorder, since many seem to act as mood stabilizers, and for the treatment of neuropathic pain. Anticonvulsants suppress the excessive rapid firing of neurons during seizures. Anticonvulsants also prevent the spread of the seizure within the brain.
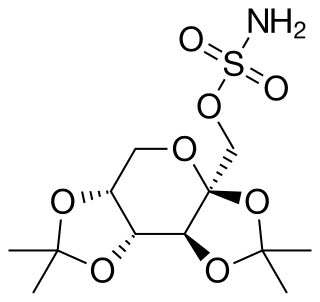
Topiramate, sold under the brand name Topamax among others, is a medication used to treat epilepsy and prevent migraines. It has also been used in alcohol dependence and essential tremor. For epilepsy this includes treatment for generalized or focal seizures. It is taken orally.

Lamotrigine, sold under the brand name Lamictal among others, is a medication used to treat epilepsy and stabilize mood in bipolar disorder. For epilepsy, this includes focal seizures, tonic-clonic seizures, and seizures in Lennox-Gastaut syndrome. In bipolar disorder, lamotrigine has not been shown to reliably treat acute depression for all groups except in the severely depressed; but for patients with bipolar disorder who are not currently symptomatic, it appears to reduce the risk of future episodes of depression.
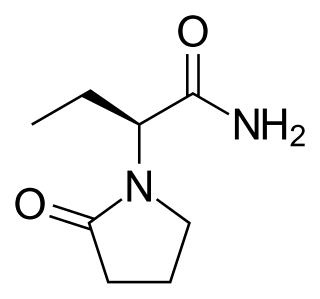
Levetiracetam, sold under the brand name Keppra among others, is a medication used to treat epilepsy. It is used for partial-onset, myoclonic, or tonic–clonic seizures and is taken either by mouth as an immediate or extended release formulation or by injection into a vein.

Viloxazine, sold under the brand name Qelbree and formerly as Vivalan among others, is a selective norepinephrine reuptake inhibitor medication which is used in the treatment of attention deficit hyperactivity disorder (ADHD) in children and adults. It was marketed for almost 30 years as an antidepressant for the treatment of depression before being discontinued and subsequently repurposed as a treatment for ADHD. Viloxazine is taken orally. It was used as an antidepressant in an immediate-release form and is used in ADHD in an extended-release form.
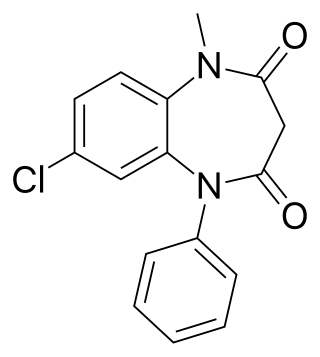
Clobazam, sold under the brand names Frisium, Onfi and others, is a benzodiazepine class medication that was patented in 1968. Clobazam was first synthesized in 1966 and first published in 1969. Clobazam was originally marketed as an anxioselective anxiolytic since 1970, and an anticonvulsant since 1984. The primary drug-development goal was to provide greater anxiolytic, anti-obsessive efficacy with fewer benzodiazepine-related side effects.

Zonisamide, sold under the brand name Zonegran among others, is a medication used to treat the symptoms of epilepsy and Parkinson's disease. Chemically it is a sulfonamide. It serves as an anticonvulsant used primarily as an adjunctive therapy in adults with Parkinson's disease, partial-onset seizures; infantile spasm, mixed seizure types of Lennox–Gastaut syndrome, myoclonic and generalized tonic clonic seizure. Despite this it is also sometimes used as a monotherapy for partial-onset seizures.
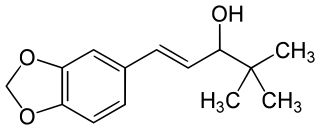
Stiripentol, sold under the brand name Diacomit, is an anticonvulsant medication used for the treatment of Dravet syndrome - a serious genetic brain disorder.
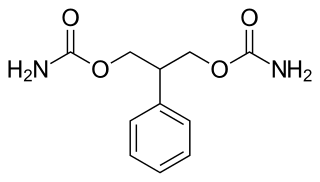
Felbamate is an anticonvulsant used in the treatment of epilepsy. It is used to treat partial seizures in adults and partial and generalized seizures associated with Lennox–Gastaut syndrome in children. However, an increased risk of potentially fatal aplastic anemia and/or liver failure limit the drug's usage to severe refractory epilepsy.

Trihexyphenidyl is an antispasmodic drug used to treat stiffness, tremors, spasms, and poor muscle control. It is an agent of the antimuscarinic class and is often used in management of Parkinson's disease. It was approved by the FDA for the treatment of Parkinson's in the US in 2003.

Tetramethylenedisulfotetramine (TETS) is an organic compound used as a rodenticide. It is an odorless, tasteless white powder that is slightly soluble in water, DMSO and acetone, and insoluble in methanol and ethanol. It is a sulfamide derivative. It can be synthesized by reacting sulfamide with formaldehyde solution in acidified water. When crystallized from acetone, it forms cubic crystals with a melting point of 255–260 °C.

Generalized epilepsy is a form of epilepsy characterised by generalised seizures with no apparent cause. Generalized seizures, as opposed to focal seizures, are a type of seizure that impairs consciousness and distorts the electrical activity of the whole or a larger portion of the brain.

Brivaracetam, sold under the brand name Briviact among others, is a chemical analog of levetiracetam, a racetam derivative with anticonvulsant (antiepileptic) properties. It is marketed by the pharmaceutical company UCB.

Lacosamide, sold under the brand name Vimpat among others, is a medication used for the treatment of partial-onset seizures and primary generalized tonic-clonic seizures. It is used by mouth or intravenously.
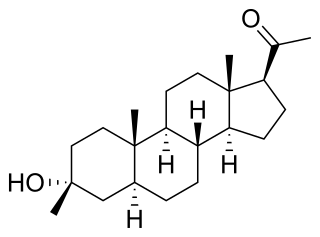
Ganaxolone, sold under the brand name Ztalmy, is a medication used to treat seizures in people with cyclin-dependent kinase-like 5 (CDKL5) deficiency disorder. Ganaxolone is a neuroactive steroid gamma-aminobutyric acid (GABA) A receptor positive modulator.

Remacemide is a drug which acts as a low-affinity NMDA antagonist with sodium channel blocking properties. It has been studied for the treatment of acute ischemic stroke, epilepsy, Huntington's disease, and Parkinson's disease.

Eslicarbazepine acetate (ESL), sold under the brand names Aptiom and Zebinix among others, is an anticonvulsant medication approved for use in Europe and the United States as monotherapy or as additional therapy for partial-onset seizures epilepsy.

Lemborexant, sold under the brand name Dayvigo, is an orexin antagonist medication which is used in the treatment of insomnia. It is indicated specifically for the treatment of insomnia characterized by difficulties with sleep onset and/or maintenance in adults. The medication is taken by mouth.

Willardiine (correctly spelled with two successive i's) or (S)-1-(2-amino-2-carboxyethyl)pyrimidine-2,4-dione is a chemical compound that occurs naturally in the seeds of Mariosousa willardiana and Acacia sensu lato. The seedlings of these plants contain enzymes capable of complex chemical substitutions that result in the formation of free amino acids (See:#Synthesis). Willardiine is frequently studied for its function in higher level plants. Additionally, many derivates of willardiine are researched for their potential in pharmaceutical development. Willardiine was first discovered in 1959 by R. Gmelin, when he isolated several free, non-protein amino acids from Acacia willardiana (another name for Mariosousa willardiana) when he was studying how these families of plants synthesize uracilyalanines. A related compound, Isowillardiine, was concurrently isolated by a different group, and it was discovered that the two compounds had different structural and functional properties. Subsequent research on willardiine has focused on the functional significance of different substitutions at the nitrogen group and the development of analogs of willardiine with different pharmacokinetic properties. In general, Willardiine is the one of the first compounds studied in which slight changes to molecular structure result in compounds with significantly different pharmacokinetic properties.
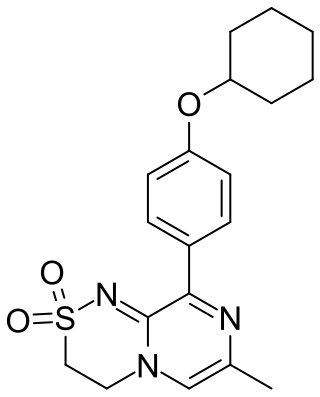
TAK-653 is an experimental drug being investigated as a treatment for treatment-resistant depression. It is being developed by Takeda Pharmaceuticals.




















