
The N-methyl-D-aspartatereceptor (also known as the NMDA receptor or NMDAR), is a glutamate receptor and ion channel found in neurons. The NMDA receptor is one of three types of ionotropic glutamate receptors, the other two being AMPA and kainate receptors. Depending on its subunit composition, its ligands are glutamate and glycine (or D-serine). However, the binding of the ligands is typically not sufficient to open the channel as it may be blocked by Mg2+ ions which are only removed when the neuron is sufficiently depolarized. Thus, the channel acts as a “coincidence detector” and only once both of these conditions are met, the channel opens and it allows positively charged ions (cations) to flow through the cell membrane. The NMDA receptor is thought to be very important for controlling synaptic plasticity and mediating learning and memory functions.

Orphenadrine is an anticholinergic drug of the ethanolamine antihistamine class; it is closely related to diphenhydramine. It is a muscle relaxant that is used to treat muscle pain and to help with motor control in Parkinson's disease, but has largely been superseded by newer drugs. It is considered a dirty drug due to its multiple mechanisms of action in different pathways. It was discovered and developed in the 1940s.

Indole alkaloids are a class of alkaloids containing a structural moiety of indole; many indole alkaloids also include isoprene groups and are thus called terpene indole or secologanin tryptamine alkaloids. Containing more than 4100 known different compounds, it is one of the largest classes of alkaloids. Many of them possess significant physiological activity and some of them are used in medicine. The amino acid tryptophan is the biochemical precursor of indole alkaloids.
Opioid-induced hyperalgesia (OIH) or opioid-induced abnormal pain sensitivity, also called paradoxical hyperalgesia, is an uncommon condition of generalized pain caused by the long-term use of high dosages of opioids such as morphine, oxycodone, and methadone. OIH is not necessarily confined to the original affected site. This means that if the person was originally taking opioids due to lower back pain, when OIH appears, the person may experience pain in the entire body, instead of just in the lower back. Over time, individuals taking opioids can also develop an increasing sensitivity to noxious stimuli, even evolving a painful response to previously non-noxious stimuli (allodynia). This means that if the person originally felt pain from twisting or from sitting too long, the person might now additionally experience pain from a light touch or from raindrops falling on the skin.

NMDA receptor antagonists are a class of drugs that work to antagonize, or inhibit the action of, the N-Methyl-D-aspartate receptor (NMDAR). They are commonly used as anesthetics for human and non-human animals; the state of anesthesia they induce is referred to as dissociative anesthesia.

Desmetramadol, also known as O-desmethyltramadol (O-DSMT), is an opioid analgesic and the main active metabolite of tramadol. Tramadol is demethylated by the liver enzyme CYP2D6 to desmetramadol in the same way as codeine, and so similarly to the variation in effects seen with codeine, individuals who have a less active form of CYP2D6 will tend to have reduced analgesic effects from tramadol. Because desmetramadol itself does not need to be metabolized to induce an analgesic effect, it can be used in individuals with low CYP2D6 activity unlike tramadol.

Psychotria colorata is a species of plant in the family Rubiaceae. It has been documented in an ethnobotanical context among the Ka'apor people of Maranhão, Brazil by Dr. William Balée, the Tulane University anthropologist and historical ecologist:

Epiboxidine is a chemical compound which acts as a partial agonist at neural nicotinic acetylcholine receptors, binding to both the α3β4 and the α4β2 subtypes. It was developed as a less toxic analogue of the potent frog-derived alkaloid epibatidine, which is around 200 times stronger than morphine as an analgesic but produces extremely dangerous toxic nicotinic side effects.
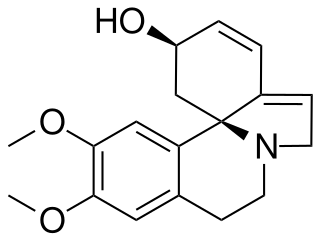
Erythravine is a tetrahydroisoquinoline alkaloid found in the plant Erythrina mulungu and other species of the genus Erythrina.
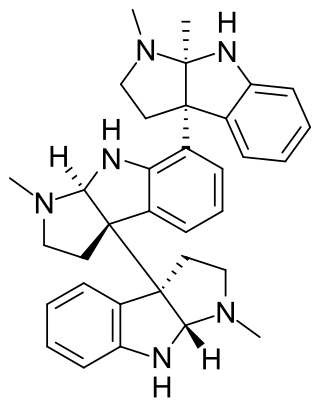
Hodgkinsine is an alkaloid found in plants of the genus Psychotria, particularly Psychotria colorata, although it is also found in Psychotria lyciiflora and probably other species in this family,

Neramexane is a drug related to memantine, which acts as an NMDA antagonist and has neuroprotective effects. It is being developed for various possible applications, including treatment of tinnitus, Alzheimer's disease, drug addiction and as an analgesic. Animal studies have also suggested antidepressant and nootropic actions, so there are a wide range of potential applications this drug may be used for. It also acts as a nicotinic acetylcholine receptor antagonist.

Arylcyclohexylamines, also known as arylcyclohexamines or arylcyclohexanamines, are a chemical class of pharmaceutical, designer, and experimental drugs.

Noribogaine, or 12-hydroxyibogamine, is the principal psychoactive metabolite of the oneirogen ibogaine. It is thought to be involved in the antiaddictive effects of ibogaine-containing plant extracts, such as Tabernanthe iboga.

Coronaridine, also known as 18-carbomethoxyibogamine, is an alkaloid found in Tabernanthe iboga and related species, including Tabernaemontana divaricata for which it was named.

ORG-25935, also known as SCH-900435 is a synthetic drug developed by Organon International, which acts as a selective inhibitor of the glycine transporter GlyT-1. In animal tests it reduces alcohol consumption and has analgesic and anticonvulsant effects, but it has mainly been studied for its antipsychotic properties, and in human trials it was shown to effectively counteract the effects of the dissociative drug ketamine.

Levomethadone, sold under the brand name L-Polamidon among others, is a synthetic opioid analgesic and antitussive which is marketed in Europe and is used for pain management and in opioid maintenance therapy. In addition to being used as a pharmaceutical drug itself, levomethadone is the main therapeutic component of methadone.

Esmethadone (INNTooltip International Nonproprietary Name; developmental code name REL-1017), also known as dextromethadone, is the (S)-enantiomer of methadone. It acts as an N-methyl-D-aspartate receptor (NMDAR) antagonist, among other actions. Unlike levomethadone, it has low affinity for opioid receptors and lacks significant respiratory depressant action and abuse liability. Esmethadone is under development for the treatment of major depressive disorder. As of August 2022, it is in phase 3 clinical trials for this indication.

Norketamine, or N-desmethylketamine, is the major active metabolite of ketamine, which is formed mainly by CYP3A4. Similarly to ketamine, norketamine acts as a noncompetitive NMDA receptor antagonist, but is about 3–5 times less potent as an anesthetic in comparison.

















