
JWH-147 is an analgesic drug used in scientific research, which acts as a cannabinoid agonist at both the CB1 and CB2 receptors. It is somewhat selective for the CB2 subtype, with a Ki of 11.0 nM at CB1 vs 7.1 nM at CB2. It was discovered and named after John W. Huffman.

JWH-307 is an analgesic drug used in scientific research, which acts as a cannabinoid agonist at both the CB1 and CB2 receptors. It is somewhat selective for the CB2 subtype, with a Ki of 7.7 nM at CB1 vs 3.3 nM at CB2. It was discovered by, and named after, John W. Huffman. JWH-307 was detected as an ingredient in synthetic cannabis smoking blends in 2012, initially in Germany.

JWH-250 or (1-pentyl-3-(2-methoxyphenylacetyl)indole) is an analgesic chemical from the phenylacetylindole family that acts as a cannabinoid agonist at both the CB1 and CB2 receptors, with a Ki of 11 nM at CB1 and 33 nM at CB2. Unlike many of the older JWH series compounds, this compound does not have a naphthalene ring, instead occupying this position with a 2'-methoxy-phenylacetyl group, making JWH-250 a representative member of a new class of cannabinoid ligands. Other 2'-substituted analogues such as the methyl, chloro and bromo compounds are also active and somewhat more potent.

JWH-210 is an analgesic chemical from the naphthoylindole family, which acts as a potent cannabinoid agonist at both the CB1 and CB2 receptors, with Ki values of 0.46 nM at CB1 and 0.69 nM at CB2. It is one of the most potent 4-substituted naphthoyl derivatives in the naphthoylindole series, having a higher binding affinity (i.e. lower Ki) at CB1 than both its 4-methyl and 4-n-propyl homologues JWH-122 (CB1 Ki 0.69 nM) and JWH-182 (CB1 Ki 0.65 nM) respectively, and than the 4-methoxy compound JWH-081 (CB1 Ki 1.2 nM). It was discovered by and named after John W. Huffman.

JWH-302 (1-pentyl-3-(3-methoxyphenylacetyl)indole) is an analgesic chemical from the phenylacetylindole family, which acts as a cannabinoid agonist with moderate affinity at both the CB1 and CB2 receptors. It is a positional isomer of the more common drug JWH-250, though it is slightly less potent with a Ki of 17 nM at CB1, compared to 11 nM for JWH-250. Because of their identical molecular weight and similar fragmentation patterns, JWH-302 and JWH-250 can be very difficult to distinguish by GC-MS testing.
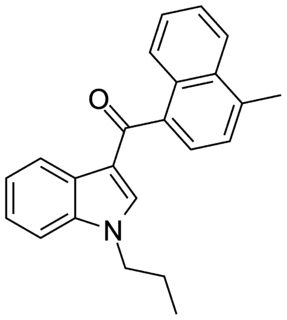
JWH-120 is a synthetic cannabimimetic that was discovered by John W. Huffman. It is the N-propyl analog of JWH-122. It is a potent and selective ligand for the CB2 receptor, but a weaker ligand for the CB1 receptor. It has a binding affinity of Ki = 6.1 ± 0.7 nM at the CB2 subtype and 173 times selectivity over the CB1 subtype.

JWH-369 ((5-(2-chlorophenyl)-1-pentyl-1H-pyrrol-3-yl)(naphthalen-1-yl)methanone) is a synthetic cannabinoid from the naphthoylpyrrole family which acts as a potent agonist of the CB1 (Ki = 7.9 ± 0.4nM) and CB2 (Ki = 5.2 ± 0.3nM) receptors, with a slight selectivity for the latter. JWH-369 was first synthesized in 2006 by John W. Huffman and colleagues to examine the nature of ligand binding to the CB1 receptor.

JWH-373 ([5-(2-butylphenyl)-1-pentylpyrrol-3-yl]-naphthalen-1-ylmethanone) is a synthetic cannabinoid from the naphthoylpyrrole family which acts as an agonist of the CB1 (Ki = 60 ± 3nM) and CB2 (Ki = 69 ± 2nM) receptors, with a slight selectivity for the former. JWH-373 was first synthesized in 2006 by John W. Huffman and colleagues to examine the nature of ligand binding to the CB1 receptor.

JWH-309 (naphthalen-1-yl-(5-naphthalen-1-yl-1-pentylpyrrol-3-yl)methanone) is a synthetic cannabinoid from the naphthoylpyrrole family which acts as an agonist of the CB1 (Ki = 41 ± 3nM) and CB2 (Ki = 49 ± 7nM) receptors, displaying a slight selectivity for the former. JWH-309 was first synthesized in 2006 by John W. Huffman and colleagues to examine the nature of ligand binding to the CB1 receptor.
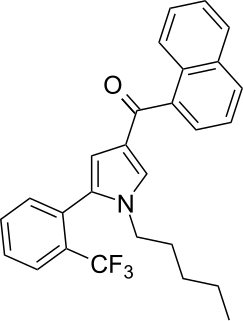
JWH-372 (naphthalen-1-yl-[1-pentyl-5-[2-(trifluoromethyl)phenyl]pyrrol-3-yl]methanone) is a synthetic cannabinoid from the naphthoylpyrrole family which acts as a potent and selective agonist of the CB2 receptor. JWH-372 binds approximately 9 times stronger to the CB2 receptor (Ki = 8.2 ± 0.2nM) than the CB1 receptor (Ki = 77 ± 2nM). The selectivity of JWH-372 for the CB2 receptor is likely due to the electron-withdrawing character of the trifluoromethyl group rather than steric effects, as the o-methyl compound JWH-370 was only mildly selective for the CB2 receptor (CB1 Ki = 5.6 ± 0.4nM, CB2 Ki = 4.0 ± 0.5nM).

JWH-371 ([5-(4-butylphenyl)-1-pentylpyrrol-3-yl]-naphthalen-1-ylmethanone) is a synthetic cannabinoid from the naphthoylpyrrole family which acts as an agonist of the CB1 (Ki = 42 ± 1nM) and CB2 (Ki = 64 ± 2nM) receptors, binding ~1.5 times stronger to the CB1 receptor than the CB2 receptor. JWH-371 was first synthesized in 2006 by John W. Huffman and colleagues to examine the nature of ligand binding to the CB1 receptor.
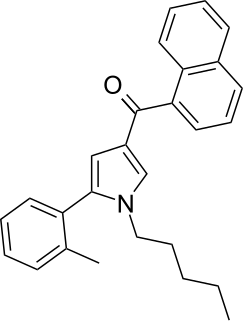
JWH-370 ([5-(2-methylphenyl)-1-pentylpyrrol-3-yl]-naphthalen-1-ylmethanone) is a synthetic cannabinoid from the naphthoylpyrrole family which acts as an agonist of the CB1 (Ki = 5.6 ± 0.4nM) and CB2 (Ki = 4.0 ± 0.5nM) receptors, with a slight selectivity for the CB2 receptor. JWH-370 was first synthesized in 2006 by John W. Huffman and colleagues to examine the nature of ligand binding to the CB1 receptor.

JWH-368 ([5-(3-Fluorophenyl)-1-pentylpyrrol-3-yl]-naphthalen-1-ylmethanone) is a synthetic cannabinoid from the naphthoylpyrrole family which acts as an agonist of the CB1 (Ki = 16 ± 1nM) and CB2 (Ki = 9.1 ± 0.7nM) receptors, binding ~1.76 times stronger to the CB2 receptor than to the CB1 receptor. JWH-368 was first synthesized in 2006 by John W. Huffman and colleagues to examine the nature of ligand binding to the CB1 receptor.

JWH-367 ([5-(3-methoxyphenyl)-1-pentylpyrrol-3-yl]-naphthalen-1-ylmethanone) is a synthetic cannabinoid from the naphthoylpyrrole family which acts as an agonist of the CB1 (Ki = 53 ± 2nM) and CB2 (Ki = 23 ± 1nM) receptors, binding ~2.3 times stronger to the CB2 receptor than to the CB1 receptor. JWH-367 was first synthesized in 2006 by John W. Huffman and colleagues to examine the nature of ligand binding to the CB1 receptor.
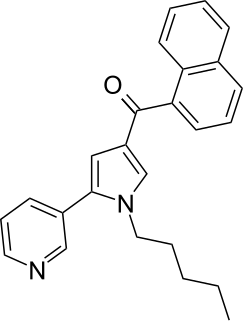
JWH-366 (naphthalen-1-yl-(1-pentyl-5-pyridin-3-ylpyrrol-3-yl)methanone) is a synthetic cannabinoid from the naphthoylpyrrole family which acts as an agonist of the CB1 (Ki = 191 ± 12nM) and CB2 (Ki = 24 ± 1nM) receptors, with a strong (~8x) selectivity for the CB2 receptor over the CB1 receptor. JWH-366 was first synthesized in 2006 by John W. Huffman and colleagues to examine the nature of ligand binding to the CB1 receptor.

JWH-365 ((5-(2-Ethylphenyl)-1-pentyl-1H-pyrrol-3-yl)(naphthalen-1-yl)methanone) is a synthetic cannabinoid from the naphthoylpyrrole family which acts as an agonist of the CB1 (Ki = 17 ± 1nM) and CB2 (Ki = 3.4 ± 0.2nM) receptors, with a strong (~5x) selectivity for the CB2 receptor over the CB1 receptor. JWH-365 was first synthesized in 2006 by John W. Huffman and colleagues to examine the nature of ligand binding to the CB1 receptor.
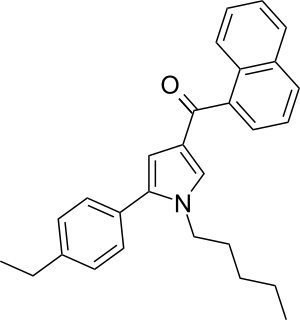
JWH-364 ([5-(4-Ethylphenyl)-1-pentyl-1H-pyrrol-3-yl](1-naphthyl)methanone) is a synthetic cannabinoid from the naphthoylpyrrole family which acts as an agonist of the CB1 (Ki = 34 ± 3nM) and CB2 (Ki = 29 ± 1nM) receptors, with a slight selectivity for the latter. JWH-364 was first synthesized in 2006 by John W. Huffman and colleagues to examine the nature of ligand binding to the CB1 receptor.

JWH-145 (1-naphthalenyl(1-pentyl-5-phenyl-1H-pyrrol-3-yl)-methanone) is a synthetic cannabinoid from the naphthoylpyrrole family which acts as an agonist of the CB1 (Ki = 14 ± 2nM) and CB2 (Ki = 6.4 ± 0.4nM) receptors, with a moderate (~2.2x) selectivity for the CB2 receptor. JWH-145 was first synthesized in 2006 by John W. Huffman and colleagues to examine the nature of ligand binding to the CB1 receptor.

JWH-146 (1-heptyl-5-phenyl-1H-pyrrol-3-yl)-1-naphthalenyl-methanone) is a synthetic cannabinoid from the naphthoylpyrrole family which acts as an agonist of the CB1 (Ki = 21 ± 2nM) and CB2 (Ki = 62 ± 5nM) receptors, with a moderate (~2.9x) selectivity for the CB1 receptor over the CB2 receptor. JWH-146 was first synthesized in 2006 by John W. Huffman and colleagues to examine the nature of ligand binding to the CB1 receptor.
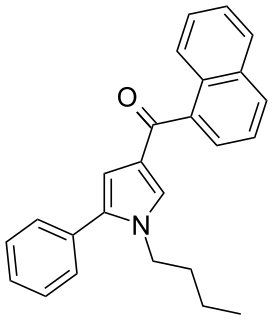
JWH-150 ((1-butyl-5-phenylpyrrol-3-yl)-naphthalen-1-ylmethanone) is a synthetic cannabinoid from the naphthoylpyrrole family which acts as an agonist of the CB1 (Ki = 60 ± 1nM) and CB2 (Ki = 15 ± 2nM) receptors, with a moderate (4x) selectivity for the CB2 receptor. JWH-150 was first synthesized in 2006 by John W. Huffman and colleagues to examine the nature of ligand binding to the CB1 receptor.




















