
APINACA (AKB48, N-(1-adamantyl)-1-pentyl-1H-indazole-3-carboxamide) is a drug that acts as a reasonably potent agonist for the cannabinoid receptors. It is a full agonist at CB1 with an EC50 of 142 nM and Ki of 3.24 nM (compared to the Ki of Δ9-THC at 28.35 nM and JWH-018 at 9.62 nM), while at CB2 it acts as a partial agonist with an EC50 of 141 nM and Ki of 1.68 nM (compared to the Ki of Δ9-THC at 37.82 nM and JWH-018 at 8.55 nM). Its pharmacological characterization has also been reported in a discontinued patent application. It had never previously been reported in the scientific or patent literature, and was first identified by laboratories in Japan in March 2012 as an ingredient in synthetic cannabis smoking blends, along with a related compound APICA. Structurally, it closely resembles cannabinoid compounds from a University of Connecticut patent, but with a simple pentyl chain on the indazole 1-position, and APINACA falls within the claims of this patent despite not being disclosed as an example.

APICA is an indole based drug that acts as a potent agonist for the cannabinoid receptors.

AB-FUBINACA (AMB-FUBINACA) is a psychoactive drug that acts as a potent agonist for the cannabinoid receptors, with Ki values of 0.9 nM at CB1 and 23.2 nM at CB2 and EC50 values of 1.8 nM at CB1 and 3.2 nM at CB2. It was originally developed by Pfizer in 2009 as an analgesic medication but was never pursued for human use. In 2012, it was discovered as an ingredient in synthetic cannabinoid blends in Japan, along with a related compound AB-PINACA, which had not previously been reported.

AB-PINACA is a compound that was first identified as a component of synthetic cannabis products in Japan in 2012.

ADBICA (also known as ADB-PICA) is a designer drug identified in synthetic cannabis blends in Japan in 2013. ADBICA had not previously been reported in the scientific literature prior to its sale as a component of synthetic cannabis blends. ADBICA features a carboxamide group at the 3-indole position, like SDB-001 and STS-135. The stereochemistry of the tert-butyl side-chain in the product is unresolved, though in a large series of indazole derivatives structurally similar to ADBICA that are disclosed in Pfizer patent WO 2009/106980, activity resides exclusively in the (S) enantiomers. ADBICA is a potent agonist of the CB1 receptor and CB2 receptor with an EC50 value of 0.69 nM and 1.8 nM respectively.

ADB-FUBINACA (ADMB-FUBINACA) is a designer drug identified in synthetic cannabis blends in Japan in 2013. In 2018, it was the third-most common synthetic cannabinoid identified in drugs seized by the Drug Enforcement Administration.
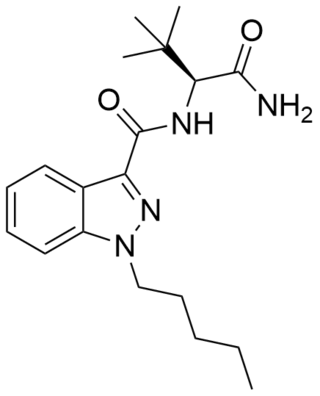
ADB-PINACA is a cannabinoid designer drug that is an ingredient in some synthetic cannabis products. It is a potent agonist of the CB1 receptor and CB2 receptor with EC50 values of 0.52 nM and 0.88 nM respectively. Like MDMB-FUBINACA, this compound incorporates the unnatural amino acid tert-leucine.

AB-CHFUPYCA is a compound that was first identified as a component of synthetic cannabis products in Japan in 2015. The name "AB-CHFUPYCA" is an acronym of its systematic name N-(1-Amino-3-methyl-1-oxoButan-2-yl)-1-(CycloHexylmethyl)-3-(4-FlUorophenyl)-1H-PYrazole-5-CarboxAmide. There are two known regioisomers of AB-CHFUPYCA: 3,5-AB-CHMFUPPYCA (pictured) and 5,3-AB-CHMFUPPYCA. The article[1] refers to both 3,5-AB-CHMFUPPYCA and 5,3-AB-CHMFUPPYCA as AB-CHMFUPPYCA isomers, so AB-CHMFUPPYCA and AB-CHFUPYCA are not names for a unique chemical structure.
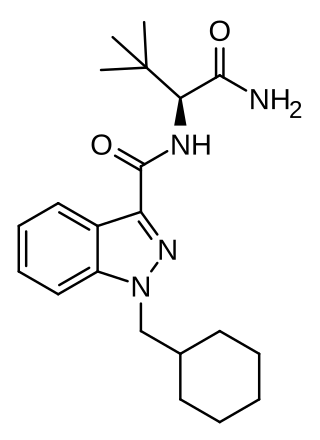
ADB-CHMINACA (also known as ADMB-CHMINACA and MAB-CHMINACA) is an indazole-based synthetic cannabinoid. It is a potent agonist of the CB1 receptor with a binding affinity of Ki = 0.289 nM and was originally developed by Pfizer in 2009 as an analgesic medication. It was identified in cannabinoid blends in Japan in early 2015.
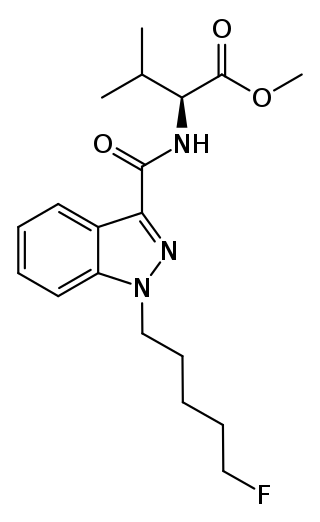
5F-AMB (also known as 5F-MMB-PINACA and 5F-AMB-PINACA) is an indazole-based synthetic cannabinoid from the indazole-3-carboxamide family, which has been used as an active ingredient in synthetic cannabis products. It was first identified in Japan in early 2014. Although only very little pharmacological information about 5F-AMB itself exists, its 4-cyanobutyl analogue (instead of 5-fluoropentyl) has been reported to be a potent agonist for the CB1 receptor (KI = 0.7 nM).

PX-3 (also known as APP-CHMINACA) is an indazole-based synthetic cannabinoid. It is a potent agonist of the CB1 receptor with a binding affinity of Ki = 47.6 nM and was originally developed by Pfizer in 2009 as an analgesic medication.

5F-AB-PINACA is an indazole-based synthetic cannabinoid that is derived from a series of compounds originally developed by Pfizer in 2009 as an analgesic medication, and has been sold online as a designer drug.
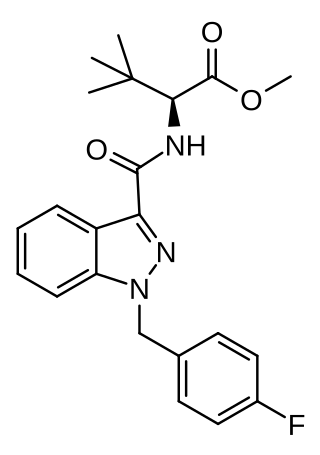
MDMB-FUBINACA (also known as MDMB(N)-Bz-F and FUB-MDMB) is an indazole-based synthetic cannabinoid that is a potent agonist for the cannabinoid receptors, with Ki values of 1.14 nM at CB1 and 0.1228 nM at CB2 and EC50 values of 0.2668 nM at CB1 and 0.1411 nM at CB2, and has been sold online as a designer drug. Its benzyl analogue (instead of 4-fluorobenzyl) has been reported to be a potent agonist for the CB1 receptor (Ki = 0.14 nM, EC50 = 2.42 nM). The structure of MDMB-FUBINACA contains the amino acid, 3-methylvaline or tert-leucine methyl ester.
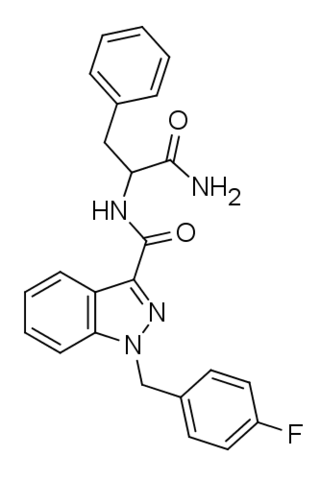
APP-FUBINACA is an indazole-based synthetic cannabinoid that has been sold online as a designer drug. Pharmacological testing showed APP-FUBINACA to have only moderate affinity for the CB1 receptor, with a Ki of 708 nM, while its EC50 was not tested. It contains a phenylalanine amino acid residue in its structure.

5F-AB-FUPPYCA (also known as AZ-037) is a pyrazole-based synthetic cannabinoid that is presumed to be an agonist of the CB1 receptor and has been sold online as a designer drug. It was first detected by the EMCDDA as part of a seizure of 540 g white powder in France in February 2015.

5F-CUMYL-P7AICA is a pyrrolo[2,3-b]pyridine-3-carboxamide based synthetic cannabinoid that has been sold as a designer drug. It was first identified by the EMCDDA in February 2015.

MDMB-4en-PINACA is an indazole-based synthetic cannabinoid that has been sold online as a designer drug. MDMB-4en-PINACA was first identified in Europe in 2017. In 2021, MDMB-4en-PINACA was the most common synthetic cannabinoid identified by the Drug Enforcement Administration in the United States. MDMB-4en-PINACA differs from 5F-MDMB-PINACA due to replacement of 5-fluoropentyl with a pent-4-ene moiety (4-en).

4F-MDMB-BINACA (also known as MDMB-4F-BINACA, 4F-MDMB-BUTINACA or 4F-ADB) is an indazole-based synthetic cannabinoid from the indazole-3-carboxamide family. It should not be confused with the amantadine analogue 4F-ABINACA. It has been used as an active ingredient in synthetic cannabis products and sold as a designer drug since late 2018. 4F-MDMB-BINACA is an agonist of the CB1 receptor (EC50 = 7.39 nM), though it is unclear whether it is selective for this target. In December 2019, the UNODC announced scheduling recommendations placing 4F-MDMB-BINACA into Schedule II throughout the world.

ADB-BINACA (also known as ADMB-BZINACA using EMCDDA naming standards) is a cannabinoid designer drug that has been found as an ingredient in some synthetic cannabis products. It was originally developed by Pfizer as a potential analgesic, and is a potent agonist of the CB1 receptor with a binding affinity (Ki) of 0.33 nM and an EC50 of 14.7 nM.

ADB-BUTINACA (also known as ADMB-BINACA using EMCDDA naming standards) is a synthetic cannabinoid compound which has been sold as a designer drug. It is a potent CB1 agonist, with a binding affinity of 0.29nM for CB1 and 0.91nM for CB2, and an EC50 of 6.36 nM for CB1.




















