
Cannabinoids are several structural classes of compounds found in the cannabis plant primarily and most animal organisms or as synthetic compounds. The most notable cannabinoid is the phytocannabinoid tetrahydrocannabinol (THC) (delta-9-THC), the primary psychoactive compound in cannabis. Cannabidiol (CBD) is also a major constituent of temperate cannabis plants and a minor constituent in tropical varieties. At least 100 distinct phytocannabinoids have been isolated from cannabis, although only four have been demonstrated to have a biogenetic origin. It was reported in 2020 that phytocannabinoids can be found in other plants such as rhododendron, licorice and liverwort, and earlier in Echinacea.

Levonantradol (CP 50,556-1) is a synthetic cannabinoid analog of dronabinol (Marinol) developed by Pfizer in the 1980s. It is around 30 times more potent than THC, and exhibits antiemetic and analgesic effects via activation of CB1 and CB2 cannabinoid receptors. Levonantradol is not currently used in medicine as dronabinol or nabilone are felt to be more useful for most conditions, however it is widely used in research into the potential therapeutic applications of cannabinoids.

Cannabinoid receptor 1 (CB1), is a G protein-coupled cannabinoid receptor that in humans is encoded by the CNR1 gene. And discovered, by determination and characterization in 1988, and cloned in 1990 for the first time. The human CB1 receptor is expressed in the peripheral nervous system and central nervous system. It is activated by endogenous cannabinoids called endocannabinoids, a group of retrograde neurotransmitters that include lipids, such as anandamide and 2-arachidonoylglycerol; plant phytocannabinoids, such as docosatetraenoylethanolamide found in wild daga, the compound tetrahydrocannabinol which is an active constituent of the psychoactive drug cannabis; and synthetic analogs of tetrahydrocannabinol. CB1 is antagonized by the phytocannabinoid tetrahydrocannabivarin at low doses and at higher doses, it activate the CB1 receptor as an agonist, but with less potency than tetrahydrocannabinol.
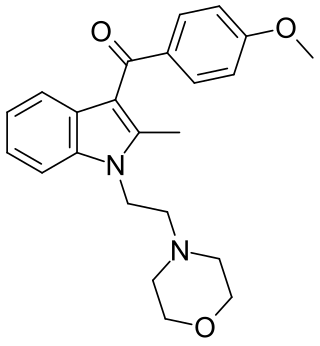
Pravadoline (WIN 48,098) is an anti-inflammatory and analgesic drug with an IC50 of 4.9 μM and a Ki of 2511 nM at CB1, related in structure to nonsteroidal anti-inflammatory drugs (NSAIDs) such as indometacin. It was developed in the 1980s as a new antiinflammatory and prostaglandin synthesis inhibitor, acting through inhibition of the enzyme cyclooxygenase (COX).
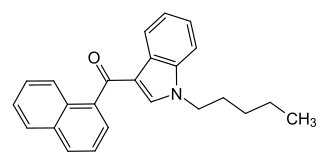
JWH-018 (1-pentyl-3-(1-naphthoyl)indole, NA-PIMO or AM-678) is an analgesic chemical from the naphthoylindole family that acts as a full agonist at both the CB1 and CB2 cannabinoid receptors, with some selectivity for CB2. It produces effects in animals similar to those of tetrahydrocannabinol (THC), a cannabinoid naturally present in cannabis, leading to its use as synthetic cannabinoid products that, in some countries, are sold legally as "incense blends".

JWH-073, a synthetic cannabinoid, is an analgesic chemical from the naphthoylindole family that acts as a full agonist at both the CB1 and CB2 cannabinoid receptors. It is somewhat selective for the CB1 subtype, with affinity at this subtype approximately 5× the affinity at CB2. The abbreviation JWH stands for John W. Huffman, one of the inventors of the compound.

CP 55,244 is a chemical compound which is a cannabinoid receptor agonist. It has analgesic effects and is used in scientific research. It is an extremely potent CB1 full agonist with a Ki of 0.21 nM, making it more potent than the commonly used full agonist HU-210.

JWH-200 (WIN 55,225) is an analgesic chemical from the aminoalkylindole family that acts as a cannabinoid receptor agonist. Its binding affinity, Ki at the CB1 receptor is 42 nM, around the same as that of THC, but its analgesic potency in vivo was higher than that of other analogues with stronger CB1 binding affinity in vitro, around 3 times that of THC but with less sedative effect, most likely reflecting favourable pharmacokinetic characteristics. It was discovered in 1991 by Sterling Drug as a potential analgesic following the earlier identification of related compounds such as pravadoline and WIN 55,212-2.

CP 47,497 or (C7)-CP 47,497 is a cannabinoid receptor agonist drug, developed by Pfizer in the 1980s. It has analgesic effects and is used in scientific research. It is a potent CB1 agonist with a Kd of 2.1 nM.

JWH-176 is an analgesic drug which acts as a cannabinoid receptor agonist. Its binding affinity at the CB1 receptor is 26.0 nM, making it more potent than THC itself, however JWH-176 is particularly notable in that it is a hydrocarbon containing no heteroatoms. This demonstrates that reasonably high-affinity cannabinoid binding and agonist effects can be produced by compounds with no hydrogen bonding capacity at all, relying merely on Van der Waals and possibly hydrophobic interactions to bind to the receptor. It was discovered by, and named after, John W. Huffman.

9-Nor-9β-hydroxyhexahydrocannabinol is a cannabinoid first discovered from early modifications to the structure of THC, in a search for the simplest compound that could still fulfill the binding requirements to produce cannabis-like activity.

Cannabicyclohexanol is a cannabinoid receptor agonist drug, developed by Pfizer in 1979. On 19 January 2009, the University of Freiburg in Germany announced that an analog of CP 47,497 was the main active ingredient in the herbal incense product Spice, specifically the 1,1-dimethyloctyl homologue of CP 47,497, which is now known as cannabicyclohexanol. The 1,1-dimethyloctyl homologue of CP 47,497 is in fact several times more potent than the parent compound, which is somewhat unexpected as the 1,1-dimethylheptyl is the most potent substituent in classical cannabinoid compounds such as HU-210.

JWH-019 is an analgesic chemical from the naphthoylindole family that acts as a cannabinoid agonist at both the CB1 and CB2 receptors. It is the N-hexyl homolog of the more common synthetic cannabinoid compound JWH-018. Unlike the butyl homolog JWH-073, which is several times weaker than JWH-018, the hexyl homolog is only slightly less potent, although extending the chain one carbon longer to the heptyl homolog JWH-020 results in dramatic loss of activity. These results show that the optimum side chain length for CB1 binding in the naphthoylindole series is the five-carbon pentyl chain, shorter than in the classical cannabinoids where a seven-carbon heptyl chain produces the most potent compounds. This difference is thought to reflect a slightly different binding conformation adopted by the naphthoylindole compounds as compared to the classical cannabinoids, and may be useful in characterizing the active site of the CB1 and CB2 receptors.

AB-001 (1-pentyl-3-(1-adamantoyl)indole) is a designer drug that was found as an ingredient in synthetic cannabis smoking blends in Ireland in 2010 and Hungary and Germany in 2011. It is unclear who AB-001 was originally developed by, but it is structurally related to compounds such as AM-1248 and its corresponding 1-(tetrahydropyran-4-ylmethyl) analogue, which are known to be potent cannabinoid agonists with moderate to a high selectivity for CB2 over CB1. The first published synthesis and pharmacological evaluation of AB-001 revealed that it acts as a full agonist at CB1 (EC50 = 35 nM) and CB2 receptors (EC50 = 48 nM). However, AB-001 was found to possess only weak cannabimimetic effects in rats at doses up to 30 mg/kg, making it less potent than the carboxamide analogue APICA, which possesses potent cannabimimetic activity at doses of 3 mg/kg.

APICA is an indole based drug that acts as a potent agonist for the cannabinoid receptors.

5F-ADB (also known as MDMB-5F-PINACA and 5F-MDMB-PINACA) is an indazole-based synthetic cannabinoid from the indazole-3-carboxamide family, which has been used as an active ingredient in synthetic cannabis products and has been sold online as a designer drug. 5F-ADB is a potent agonist of the CB1 receptor, though it is unclear whether it is selective for this target. 5F-ADB was first identified in November 2014 from post-mortem samples taken from an individual who had died after using a product containing this substance. Subsequent testing identified 5F-ADB to have been present in a total of ten people who had died from unexplained drug overdoses in Japan between September 2014 and December 2014. 5F-ADB is believed to be extremely potent based on the very low levels detected in tissue samples, and appears to be significantly more toxic than earlier synthetic cannabinoid drugs that had previously been sold.
Guineesine is a compound isolated from long pepper and black pepper.
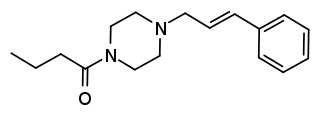
Bucinnazine is an opioid analgesic drug that was widely used in China to treat pain in cancer patients as of 1986. It is one of the most potent compounds among a series of piperazine-amides first synthesized and reported in Japan in the 1970s. Bucinnazine has analgesic potency comparable to that of morphine, but with a relatively higher therapeutic index.

















