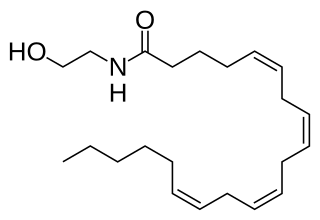
Anandamide (ANA), also referred to as N-arachidonoylethanolamine (AEA) is a fatty acid neurotransmitter belonging to the fatty acid derivative group known as N-acylethanolamine (NAE). Anandamide takes its name from the Sanskrit word ananda, meaning "joy, bliss, delight," plus amide. Anandamide, the first discovered endocannabinoid, engages with the body's endocannabinoid system by binding to the same cannabinoid receptors that THC found in cannabis acts on. Anandamide can be found within tissues in a wide range of animals. It has also been found in plants, such as the cacao tree.

Cannabinoids are several structural classes of compounds found in the cannabis plant primarily and most animal organisms or as synthetic compounds. The most notable cannabinoid is the phytocannabinoid tetrahydrocannabinol (THC) (delta-9-THC), the primary psychoactive compound in cannabis. Cannabidiol (CBD) is also a major constituent of temperate cannabis plants and a minor constituent in tropical varieties. At least 100 distinct phytocannabinoids have been isolated from cannabis, although only four have been demonstrated to have a biogenetic origin. It was reported in 2020 that phytocannabinoids can be found in other plants such as rhododendron, licorice and liverwort, and earlier in Echinacea.

Cannabinoid receptors, located throughout the body, are part of the endocannabinoid system of vertebrates– a class of cell membrane receptors in the G protein-coupled receptor superfamily. As is typical of G protein-coupled receptors, the cannabinoid receptors contain seven transmembrane spanning domains. Cannabinoid receptors are activated by three major groups of ligands:

HU-210 is a synthetic cannabinoid that was first synthesized in 1988 from (1R,5S)-myrtenol by a group led by Raphael Mechoulam at the Hebrew University. HU-210 is 100 to 800 times more potent than natural THC from cannabis and has an extended duration of action. HU-210 has a binding affinity of 0.061 nM at CB1 receptors compared to 40.7 nM for Δ9-THC. The binding pose of HU-210 to the CB1 receptor is similar to other synthetic cannabinoids.

Levonantradol (CP 50,556-1) is a synthetic cannabinoid analog of dronabinol (Marinol) developed by Pfizer in the 1980s. It is around 30 times more potent than THC, and exhibits antiemetic and analgesic effects via activation of CB1 and CB2 cannabinoid receptors. Levonantradol is not currently used in medicine as dronabinol or nabilone are felt to be more useful for most conditions, however it is widely used in research into the potential therapeutic applications of cannabinoids.

Nabitan (nabutam, benzopyranoperidine, SP-106, Abbott 40656) is a synthetic cannabinoid analog of dronabinol (Δ9-tetrahydrocannabinol) and dimethylheptylpyran. It exhibits antiemetic and analgesic effects, most likely by binding to and activating the CB1 and CB2 cannabinoid receptors, and reduced intraocular pressure in animal tests, making it potentially useful in the treatment of glaucoma.

Dimethylheptylpyran (DMHP) is a synthetic cannabinoid and analogue of tetrahydrocannabinol (THC). It was invented in 1949 during attempts to elucidate the structure of Δ9-THC, one of the active components of cannabis. DMHP is a pale yellow, viscous oil which is insoluble in water but dissolves in alcohol or non-polar solvents.
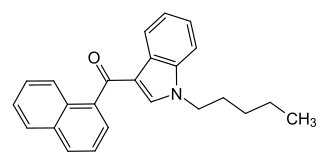
JWH-018 (1-pentyl-3-(1-naphthoyl)indole, NA-PIMO or AM-678) is an analgesic chemical from the naphthoylindole family that acts as a full agonist at both the CB1 and CB2 cannabinoid receptors, with some selectivity for CB2. It produces effects in animals similar to those of tetrahydrocannabinol (THC), a cannabinoid naturally present in cannabis, leading to its use as synthetic cannabinoid products that, in some countries, are sold legally as "incense blends".
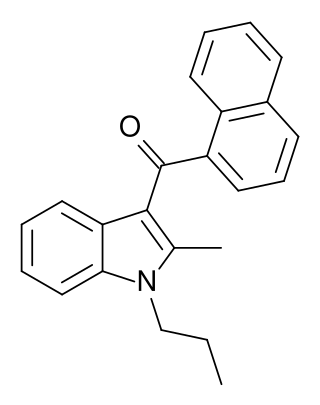
JWH-015 is a chemical from the naphthoylindole family that acts as a subtype-selective cannabinoid agonist. Its affinity for CB2 receptors is 13.8 nM, while its affinity for CB1 is 383 nM, meaning that it binds almost 28 times more strongly to CB2 than to CB1. However, it still displays some CB1 activity, and in some model systems can be very potent and efficacious at activating CB1 receptors, and therefore it is not as selective as newer drugs such as JWH-133. It has been shown to possess immunomodulatory effects, and CB2 agonists may be useful in the treatment of pain and inflammation. It was discovered and named after John W. Huffman.

Surinabant (SR147778) is a cannabinoid receptor type 1 antagonist developed by Sanofi-Aventis. It is being investigated as a potential treatment for nicotine addiction, to assist smoking cessation. It may also be developed as an anorectic drug to assist with weight loss, however there are already several CB1 antagonists or inverse agonists on the market or under development for this application, so surinabant is at present mainly being developed as an anti-smoking drug, with possible application in the treatment of other addictive disorders such as alcoholism. Other potential applications such as treatment of ADHD have also been proposed.

Synthetic cannabinoids are a class of designer drug molecules that bind to the same receptors to which cannabinoids in cannabis plants attach. These novel psychoactive substances should not be confused with synthetic phytocannabinoids or synthetic endocannabinoids from which they are in many aspects distinct.

Cannabicyclohexanol is a cannabinoid receptor agonist drug, developed by Pfizer in 1979. On 19 January 2009, the University of Freiburg in Germany announced that an analog of CP 47,497 was the main active ingredient in the herbal incense product Spice, specifically the 1,1-dimethyloctyl homologue of CP 47,497, which is now known as cannabicyclohexanol. The 1,1-dimethyloctyl homologue of CP 47,497 is in fact several times more potent than the parent compound, which is somewhat unexpected as the 1,1-dimethylheptyl is the most potent substituent in classical cannabinoid compounds such as HU-210.

AM-2233 is a drug that acts as a highly potent full agonist for the cannabinoid receptors, with a Ki of 1.8 nM at CB1 and 2.2 nM at CB2 as the active (R) enantiomer. It was developed as a selective radioligand for the cannabinoid receptors and has been used as its 131I derivative for mapping the distribution of the CB1 receptor in the brain. AM-2233 was found to fully substitute for THC in rats, with a potency lower than that of JWH-018 but higher than WIN 55,212-2.
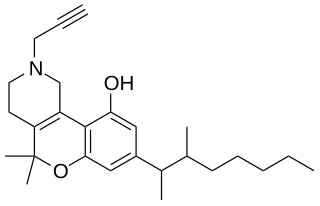
A-40174 (SP-1) is an analgesic drug which acts as a potent cannabinoid receptor agonist, and was developed by Abbott Laboratories in the 1970s. It's a structural analog of dronabinol and dimethylheptylpyran.

Menabitan is a synthetic drug which acts as a potent cannabinoid receptor agonist. It is closely related to natural cannabinoids of the tetrahydrocannabinol (THC) group, differing mainly by its longer and branched side chain, and the replacement of the 9-position carbon with a nitrogen. It's a structural analog of nabitan and dimethylheptylpyran. It was studied as an analgesic in the 1970s and was found to possess analgesic effects in both humans and animals but was never marketed.
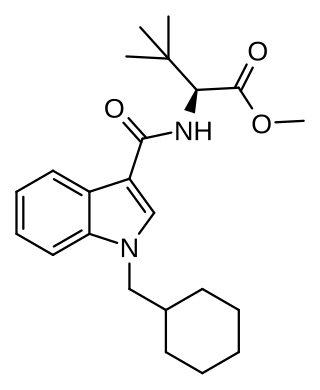
'MDMB-CHMICAa' is an indole-based synthetic cannabinoid that is a potent agonist of the CB1 receptor and has been sold online as a designer drug. While MDMB-CHMICA was initially sold under the name "MMB-CHMINACA", the compound corresponding to this code name (i.e. the isopropyl instead of t-butyl analogue of MDMB-CHMINACA) has been identified on the designer drug market in 2015 as AMB-CHMINACA.

5F-MDMB-PICA (MDMB-5F-PICA) is a designer drug and synthetic cannabinoid. In 2018, it was the fifth-most common synthetic cannabinoid identified in drugs seized by the Drug Enforcement Administration.

MDMB-4en-PINACA is an indazole-based synthetic cannabinoid that has been sold online as a designer drug. MDMB-4en-PINACA was first identified in Europe in 2017. In 2021, MDMB-4en-PINACA was the most common synthetic cannabinoid identified by the Drug Enforcement Administration in the United States. MDMB-4en-PINACA differs from 5F-MDMB-PINACA due to replacement of 5-fluoropentyl with a pent-4-ene moiety (4-en).

CP 42,096 is an analgesic drug which acts as a cannabinoid agonist. It was developed by Pfizer in the 1980s as part of the research that led to the development of levonantradol, and is more potent than THC but less potent than newer compounds such as CP 55,244.



















