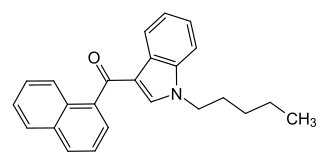
JWH-018 (1-pentyl-3-(1-naphthoyl)indole, NA-PIMO or AM-678) is an analgesic chemical from the naphthoylindole family that acts as a full agonist at both the CB1 and CB2 cannabinoid receptors, with some selectivity for CB2. It produces effects in animals similar to those of tetrahydrocannabinol (THC), a cannabinoid naturally present in cannabis, leading to its use as synthetic cannabinoid products that, in some countries, are sold legally as "incense blends".

JWH-081 is an analgesic chemical from the naphthoylindole family, which acts as a cannabinoid agonist at both the CB1 and CB2 receptors. With a Ki of 1.2nM it is fairly selective for the CB1 subtype, its affinity at this subtype is measured at approximately 10x the affinity at CB2(12.4nM). It was discovered by and named after John W. Huffman.

JWH-073, a synthetic cannabinoid, is an analgesic chemical from the naphthoylindole family that acts as a full agonist at both the CB1 and CB2 cannabinoid receptors. It is somewhat selective for the CB1 subtype, with affinity at this subtype approximately 5× the affinity at CB2. The abbreviation JWH stands for John W. Huffman, one of the inventors of the compound.
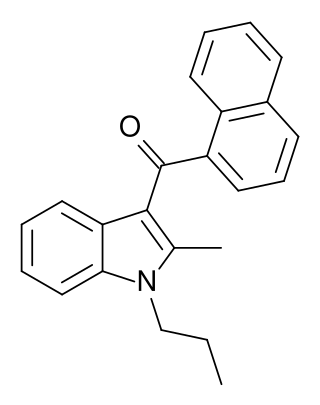
JWH-015 is a chemical from the naphthoylindole family that acts as a subtype-selective cannabinoid agonist. Its affinity for CB2 receptors is 13.8 nM, while its affinity for CB1 is 383 nM, meaning that it binds almost 28 times more strongly to CB2 than to CB1. However, it still displays some CB1 activity, and in some model systems can be very potent and efficacious at activating CB1 receptors, and therefore it is not as selective as newer drugs such as JWH-133. It has been shown to possess immunomodulatory effects, and CB2 agonists may be useful in the treatment of pain and inflammation. It was discovered and named after John W. Huffman.

JWH-250 or (1-pentyl-3-(2-methoxyphenylacetyl)indole) is an analgesic chemical from the phenylacetylindole family that acts as a cannabinoid agonist at both the CB1 and CB2 receptors, with a Ki of 11 nM at CB1 and 33 nM at CB2. Unlike many of the older JWH series compounds, this compound does not have a naphthalene ring, instead occupying this position with a 2'-methoxy-phenylacetyl group, making JWH-250 a representative member of a new class of cannabinoid ligands. Other 2'-substituted analogues such as the methyl, chloro and bromo compounds are also active and somewhat more potent.

JWH-359 is a dibenzopyran "classical" cannabinoid drug, which is a potent and selective CB2 receptor agonist, with a Ki of 13.0 nM and selectivity of around 220 times for CB2 over CB1 receptors. It is related to other dibenzopyran CB2 agonists such as JWH-133 and L-759,656 but with a chiral side chain which has made it useful for mapping the shape of the CB2 binding site. It was discovered by, and named after, John W. Huffman.

JWH-203 (1-pentyl-3-(2-chlorophenylacetyl)indole) is an analgesic chemical from the phenylacetylindole family that acts as a cannabinoid agonist with approximately equal affinity at both the CB1 and CB2 receptors, having a Ki of 8.0 nM at CB1 and 7.0 nM at CB2. It was originally discovered by, and named after, John W. Huffman, but has subsequently been sold without his permission as an ingredient of synthetic cannabis smoking blends. Similar to the related 2'-methoxy compound JWH-250, the 2'-bromo compound JWH-249, and the 2'-methyl compound JWH-251, JWH-203 has a phenylacetyl group in place of the naphthoyl ring used in most aminoalkylindole cannabinoid compounds, and has the strongest in vitro binding affinity for the cannabinoid receptors of any compound in the phenylacetyl group.

JWH-210 is an analgesic chemical from the naphthoylindole family, which acts as a potent cannabinoid agonist at both the CB1 and CB2 receptors, with Ki values of 0.46 nM at CB1 and 0.69 nM at CB2. It is one of the most potent 4-substituted naphthoyl derivatives in the naphthoylindole series, having a higher binding affinity (i.e. lower Ki) at CB1 than both its 4-methyl and 4-n-propyl homologues JWH-122 (CB1 Ki 0.69 nM) and JWH-182 (CB1 Ki 0.65 nM) respectively, and than the 4-methoxy compound JWH-081 (CB1 Ki 1.2 nM). It was discovered by and named after John W. Huffman.

JWH-007 is an analgesic chemical from the naphthoylindole family, which acts as a cannabinoid agonist at both the CB1 and CB2 receptors. It was first reported in 1994 by a group including the noted cannabinoid chemist John W. Huffman. It was the most active of the first group of N-alkyl naphoylindoles discovered by the team led by John W Huffman, several years after the family was initially described with the discovery of the N-morpholinylethyl compounds pravadoline (WIN 48,098), JWH-200 (WIN 55,225) and WIN 55,212-2 by the Sterling Winthrop group. Several other N-alkyl substituents were found to be active by Huffman's team including the n-butyl, n-hexyl, 2-heptyl, and cyclohexylethyl groups, but it was subsequently determined that the 2-methyl group on the indole ring is not required for CB1 binding, and tends to increase affinity for CB2 instead. Consequently, the 2-desmethyl derivative of JWH-007, JWH-018, has slightly higher binding affinity for CB1, with an optimum binding of 9.00 nM at CB1 and 2.94 nM at CB2, and JWH-007 displayed optimum binding of 9.50 nM at CB1 and 2.94 nM at CB2.
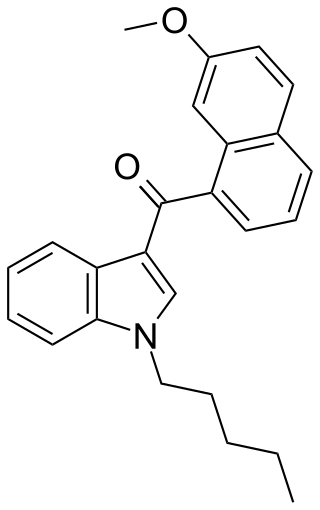
JWH-164 is a synthetic cannabinoid receptor agonist from the naphthoylindole family. It has approximately equal affinity for the CB1 and CB2 receptors, with a Ki of 6.6 nM at CB1 and 6.9 nM at CB2. JWH-164 is a positional isomer of the related compound JWH-081, but with a methoxy group at the 7-position of the naphthyl ring, rather than the 4-position as in JWH-081. Its potency is intermediate between that of JWH-081 and its ring unsubstituted derivative JWH-018, demonstrating that substitution of the naphthyl 7-position can also result in increased cannabinoid receptor binding affinity.
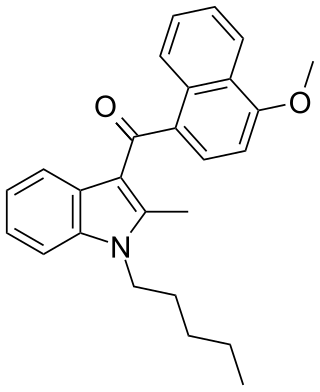
JWH-098 is a synthetic cannabinoid receptor agonist from the naphthoylindole family. It is the indole 2-methyl derivative of a closely related compound JWH-081, but has markedly different affinity for the CB1 and CB2 receptors. While JWH-081 is around tenfold selective for CB1 over CB2, in JWH-098 this is reversed, and it is around four times weaker than JWH-081 at CB1 while being six times more potent at CB2, giving it a slight selectivity for CB2 overall. This makes JWH-098 a good example of how methylation of the indole 2-position in the naphthoylindole series tends to increase CB2 affinity, but often at the expense of CB1 binding.

A-834,735 is a drug developed by Abbott Laboratories that acts as a potent cannabinoid receptor full agonist at both the CB1 and CB2 receptors, with a Ki of 12 nM at CB1 and 0.21 nM at CB2. Replacing the aromatic 3-benzoyl or 3-naphthoyl group found in most indole derived cannabinoids with the 3-tetramethylcyclopropylmethanone group of A-834,735 and related compounds imparts significant selectivity for CB2, with most compounds from this group found to be highly selective CB2 agonists with little affinity for CB1. However, low nanomolar CB1 binding affinity is retained with certain heterocyclic 1-position substituents such as (N-methylpiperidin-2-yl)methyl (cf. AM-1220, AM-1248), or the (tetrahydropyran-4-yl)methyl substituent of A-834,735, resulting in compounds that still show significant affinity and efficacy at both receptors despite being CB2 selective overall.

JWH-175 is a drug from the naphthylmethylindole family which acts as a cannabinoid receptor agonist. It was invented by the scientist John W. Huffman and colleagues at Clemson University. JWH-175 is closely related to the widely used cannabinoid designer drug JWH-018, but with the ketone bridge replaced by a simpler methylene bridge. It is several times weaker than JWH-018, having a binding affinity at the CB1 receptor of 22 nM, though some derivatives substituted at the 4-position of the naphthyl ring have potency more closely approaching that of the equivalent naphthoylindoles. This makes JWH-175 considerably less potent than most synthetic cannabinoid drugs used in synthetic cannabis blends, and it is unclear if JWH-175 has ever been used for this purpose. However it has still been explicitly banned in several jurisdictions including Russia and some Australian states, in order to stop its potential use as an ingredient in such products. In the United States, all CB1 receptor agonists of the 3-(1-naphthylmethane)indole class such as JWH-175 are Schedule I Controlled Substances.

JWH-047 is a selective cannabinoid ligand that binds to both CB1 and CB2. It has a bindining affinity of Ki = 0.9 nM for the CB2 subtype, and more than 65 times selectivity over the CB1.
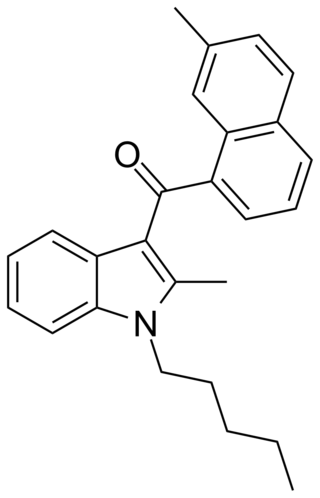
JWH-048 is a selective cannabinoid ligand, with a bindining affinity of Ki = 0.5 ± 0.1 nM for the CB2 subtype, and more than 22 times selectivity over the CB1.
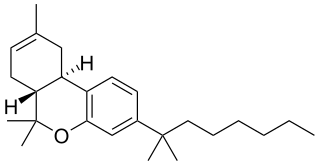
JWH-057, also known as deoxy-Δ8-THC-DMH, is a selective cannabinoid ligand, with a binding affinity of Ki = 2.9 ± 1.6 nM for the CB2 subtype, and Ki = 23 ± 7 nM for CB1.

JWH-116 is a synthetic cannabinoid receptor ligand from the naphthoylindole family. It is the indole 2-ethyl derivative of related compound JWH-018. The binding affinity of JWH-116 for the CB1 receptor is reported as Ki = 52 ± 5 nM.
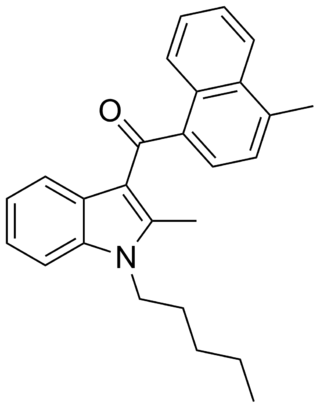
JWH-149 is a synthetic cannabimimetic that was discovered by John W. Huffman. It is the N-pentyl analog of JWH-148. It is a potent but only moderately selective ligand for the CB2 receptor, with a binding affinity of Ki = 0.73 ± 0.03 nM at this subtype, and more than six times selectivity over the CB1 subtype.

Tetrahydrocannabiphorol (THCP) is a potent phytocannabinoid, a CB1 and CB2 receptor agonist which was known as a synthetic homologue of tetrahydrocannabinol (THC), but for the first time in 2019 was isolated as a natural product in trace amounts from Cannabis sativa.

JWH-146 (1-heptyl-5-phenyl-1H-pyrrol-3-yl)-1-naphthalenyl-methanone) is a synthetic cannabinoid from the naphthoylpyrrole family which acts as an agonist of the CB1 (Ki = 21 ± 2nM) and CB2 (Ki = 62 ± 5nM) receptors, with a moderate (~2.9x) selectivity for the CB1 receptor over the CB2 receptor. JWH-146 was first synthesized in 2006 by John W. Huffman and colleagues to examine the nature of ligand binding to the CB1 receptor.




















