 | |
 | |
| Names | |
|---|---|
| Other names octadecaborane; octadecaboron doicosahydride; octodecaborane; n-Octadecaborane; i-Octadecaborane | |
| Identifiers | |
3D model (JSmol) | |
| ECHA InfoCard | 100.224.871 |
| EC Number |
|
CompTox Dashboard (EPA) | |
| |
| |
| Properties | |
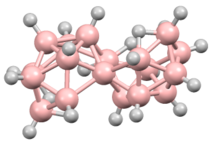
| B18H22 | |
| Molar mass | 216.77 g/mol |
| Appearance | White to off white powder |
| Density | 1.012 g/cm3 |
| Melting point | 180 and 129 °C (356 and 264 °F; 453 and 402 K) n-B18H22 and i-B18H22 respectively |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
Octadecaborane is an inorganic compound, a boron hydride cluster with chemical formula B18H22. It is a colorless flammable solid, like many higher boron hydrides. Although the compound has no practical applications, its structure is of theoretical and pedagogical interest.