
Tyrosine-protein kinase ABL1 also known as ABL1 is a protein that, in humans, is encoded by the ABL1 gene located on chromosome 9. c-Abl is sometimes used to refer to the version of the gene found within the mammalian genome, while v-Abl refers to the viral gene, which was initially isolated from the Abelson murine leukemia virus.

Growth factor receptor-bound protein 2, also known as Grb2, is an adaptor protein involved in signal transduction/cell communication. In humans, the GRB2 protein is encoded by the GRB2 gene.

Tyrosine-protein phosphatase non-receptor type 11 (PTPN11) also known as protein-tyrosine phosphatase 1D (PTP-1D), Src homology region 2 domain-containing phosphatase-2 (SHP-2), or protein-tyrosine phosphatase 2C (PTP-2C) is an enzyme that in humans is encoded by the PTPN11 gene. PTPN11 is a protein tyrosine phosphatase (PTP) Shp2.

Adapter molecule crk also known as proto-oncogene c-Crk is a protein that in humans is encoded by the CRK gene.

Growth factor receptor-bound protein 10 also known as insulin receptor-binding protein Grb-IR is a protein that in humans is encoded by the GRB10 gene.

Proto-oncogene tyrosine-protein kinase Fyn is an enzyme that in humans is encoded by the FYN gene.

Phosphatidylinositol 3-kinase regulatory subunit alpha is an enzyme that in humans is encoded by the PIK3R1 gene.
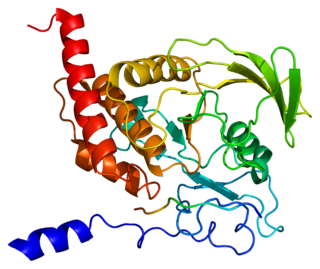
Tyrosine-protein phosphatase non-receptor type 6, also known as Src homology region 2 domain-containing phosphatase-1 (SHP-1), is an enzyme that in humans is encoded by the PTPN6 gene.
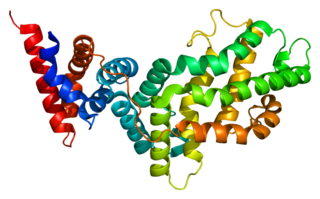
RAS p21 protein activator 1 or RasGAP, also known as RASA1, is a 120-kDa cytosolic human protein that provides two principal activities:

GRB2-associated-binding protein 1 is a protein that in humans is encoded by the GAB1 gene.

Protein tyrosine kinase 2 beta is an enzyme that in humans is encoded by the PTK2B gene.

CBL-B is an E3 ubiquitin-protein ligase that in humans is encoded by the CBLB gene. CBLB is a member of the CBL gene family.

Fibroblast growth factor receptor substrate 2 is a protein that in humans is encoded by the FRS2 gene.

Proto-oncogene vav is a protein that in humans is encoded by the VAV1 gene.
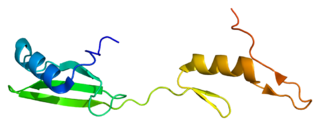
Cytoplasmic protein NCK1 is a protein that in humans is encoded by the NCK1 gene.

Tyrosine-protein phosphatase non-receptor type 12 is an enzyme that in humans is encoded by the PTPN12 gene.

BAG family molecular chaperone regulator 3 is a protein that in humans is encoded by the BAG3 gene. BAG3 is involved in chaperone-assisted selective autophagy.

SH2B adapter protein 2 is a protein that in humans is encoded by the SH2B2 gene.

Tyrosine-protein kinase BLK, also known as B lymphocyte kinase, is a non-receptor tyrosine kinase that in humans is encoded by the BLK gene. It is of the Src family of tyrosine kinases.

Signal transduction protein CBL-C is a protein that in humans is encoded by the CBLC gene.






























