Related Research Articles

Curium is a synthetic chemical element; it has symbol Cm and atomic number 96. This transuranic actinide element was named after eminent scientists Marie and Pierre Curie, both known for their research on radioactivity. Curium was first intentionally made by the team of Glenn T. Seaborg, Ralph A. James, and Albert Ghiorso in 1944, using the cyclotron at Berkeley. They bombarded the newly discovered element plutonium with alpha particles. This was then sent to the Metallurgical Laboratory at University of Chicago where a tiny sample of curium was eventually separated and identified. The discovery was kept secret until after the end of World War II. The news was released to the public in November 1947. Most curium is produced by bombarding uranium or plutonium with neutrons in nuclear reactors – one tonne of spent nuclear fuel contains ~20 grams of curium.
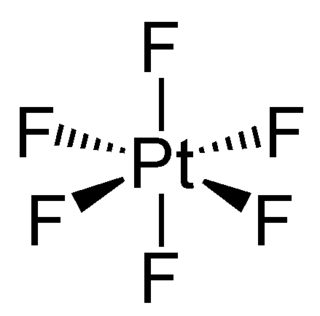
Platinum hexafluoride is the chemical compound with the formula PtF6, and is one of seventeen known binary hexafluorides. It is a dark-red volatile solid that forms a red gas. The compound is a unique example of platinum in the +6 oxidation state. With only four d-electrons, it is paramagnetic with a triplet ground state. PtF6 is a strong fluorinating agent and one of the strongest oxidants, capable of oxidising xenon and O2. PtF6 is octahedral in both the solid state and in the gaseous state. The Pt-F bond lengths are 185 picometers.

Curium(III) oxide is a compound composed of curium and oxygen with the chemical formula Cm2O3. It is a crystalline solid with a unit cell that contains two curium atoms and three oxygen atoms. The simplest synthesis equation involves the reaction of curium(III) metal with O2−: 2 Cm3+ + 3 O2− ---> Cm2O3. Curium trioxide can exist as five polymorphic forms. Two of the forms exist at extremely high temperatures, making it difficult for experimental studies to be done on the formation of their structures. The three other possible forms which curium sesquioxide can take are the body-centered cubic form, the monoclinic form, and the hexagonal form. Curium(III) oxide is either white or light tan in color and, while insoluble in water, is soluble in inorganic and mineral acids. Its synthesis was first recognized in 1955.

Technetium hexafluoride or technetium(VI) fluoride (TcF6) is a yellow inorganic compound with a low melting point. It was first identified in 1961. In this compound, technetium has an oxidation state of +6, the highest oxidation state found in the technetium halides. In this respect, technetium differs from rhenium, which forms a heptafluoride, ReF7. Technetium hexafluoride occurs as an impurity in uranium hexafluoride, as technetium is a fission product of uranium (spontaneous fission in natural uranium, possible contamination from induced fission inside the reactor in reprocessed uranium). The fact that the boiling point of the hexafluorides of uranium and technetium are very close to each other presents a problem in using fluoride volatility in nuclear reprocessing.
Selenium hexafluoride is the inorganic compound with the formula SeF6. It is a very toxic colourless gas described as having a "repulsive" odor. It is not widely encountered and has no commercial applications.
A hexafluoride is a chemical compound with the general formula QXnF6, QXnF6m−, or QXnF6m+. Many molecules fit this formula. An important hexafluoride is hexafluorosilicic acid (H2SiF6), which is a byproduct of the mining of phosphate rock. In the nuclear industry, uranium hexafluoride (UF6) is an important intermediate in the purification of this element.

Iridium hexafluoride, also iridium(VI) fluoride, (IrF6) is a compound of iridium and fluorine and one of the seventeen known binary hexafluorides. It is one of only a few compounds with iridium in the oxidation state +6.

Molybdenum hexafluoride, also molybdenum(VI) fluoride, is the inorganic compound with the formula MoF6. It is the highest fluoride of molybdenum. It is a colourless solid and melts just below room temperature and boils in 34 °C. It is one of the seventeen known binary hexafluorides.

Curium hydroxideCm(OH)3 is a radioactive compound first discovered in measurable quantities in 1947. It is composed of a single curium atom and three hydroxy groups. It was the first curium compound ever isolated.

Rhenium hexafluoride, also rhenium(VI) fluoride, (ReF6) is a compound of rhenium and fluorine and one of the seventeen known binary hexafluorides.

Curium(III) fluoride or curium trifluoride is the chemical compound composed of curium and fluorine with the formula CmF3. It is a white, nearly insoluble salt that has the same crystal structure as LaF3. It precipitates as a hydrate when fluoride ions are added to a weakly acidic Cm(III) solution; alternatively it can be synthesized by reacting hydrofluoric acid with Cm(OH)3. The anhydrous form is then obtained by desiccation or by treatment with hydrogen fluoride gas.
Curium(III) nitrate is an inorganic compound, a salt of curium and nitric acid with the chemical formula Cm(NO3)3.
Curium compounds are compounds containing the element curium (Cm). Curium usually forms compounds in the +3 oxidation state, although compounds with curium in the +4, +5 and +6 oxidation states are also known.

Curium(III) chloride is the chemical compound with the formula CmCl3.
Palladium hexafluoride is an inorganic chemical compound of palladium metal and fluorine with the chemical formula PdF6. It is reported to be a still hypothetical compound. This is one of many palladium fluorides.

Curium(IV) fluoride is an inorganic chemical compound, a salt of curium and fluorine with the chemical formula CmF4.

Einsteinium hexafluoride is a binary inorganic chemical compound of einsteinium and fluorine with the chemical formula EsF6. This is a hypothetical compound—its existence has been predicted theoretically, but the compound has yet to be isolated.

Curium(IV) oxide is an inorganic chemical compound of curium and oxygen with the chemical formula CmO2. Since all isotopes of curium are man-made, the compound does not occur in nature.

Curium(III) iodide is the chemical compound with the formula CmI3. Since all isotopes of curium are only artificially produced, the compound has no natural occurrence.
Curium fluoride may refer to:
References
- ↑ Seppelt, Konrad (28 January 2015). "Molecular Hexafluorides". Chemical Reviews . 115 (2): 1296–1306. doi:10.1021/cr5001783. PMID 25418862 . Retrieved 27 June 2023.
- 1 2 Macintyre, Jane E. (23 July 1992). Dictionary of Inorganic Compounds. CRC Press. p. 3046. ISBN 978-0-412-30120-9 . Retrieved 27 June 2023.