 | |
 Osmium dioxide dihydrate | |
| Names | |
|---|---|
| IUPAC name Osmium dioxide | |
| Other names Osmium(IV) oxide | |
| Identifiers | |
3D model (JSmol) | |
| ChemSpider | |
PubChem CID | |
CompTox Dashboard (EPA) | |
| |
| |
| Properties | |
| OsO2 | |
| Molar mass | 222.229 g/mol |
| Appearance | black or yellow brown |
| Density | 11.4 g/cm3 |
| Melting point | 500 °C (932 °F; 773 K) (decomposes) |
| insoluble [1] | |
| Solubility | dissolves in HCl |
| Related compounds [2] | |
| Osmium tetroxide | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
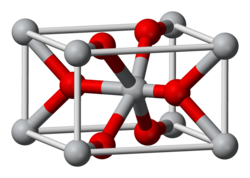
Osmium dioxide is an inorganic compound with the formula OsO2. It exists as brown to black crystalline powder, but single crystals are golden and exhibit metallic conductivity. The compound crystallizes in the rutile structural motif, i.e. the connectivity is very similar to that in the mineral rutile.