
Heroin, also known as diacetylmorphine and diamorphine among other names, is a morphinan opioid substance synthesized from the dried latex of the opium poppy; it is mainly used as a recreational drug for its euphoric effects. Heroin is used medically in several countries to relieve pain, such as during childbirth or a heart attack, as well as in opioid replacement therapy. Medical-grade diamorphine is used as a pure hydrochloride salt. Various white and brown powders sold illegally around the world as heroin are routinely diluted with cutting agents. Black tar heroin is a variable admixture of morphine derivatives—predominantly 6-MAM (6-monoacetylmorphine), which is the result of crude acetylation during clandestine production of street heroin.

Morphine, formerly also called morphia, is an opiate that is found naturally in opium, a dark brown resin produced by drying the latex of opium poppies. It is mainly used as an analgesic. There are numerous methods used to administer morphine: oral; sublingual; via inhalation; injection into a muscle, injection under the skin, or injection into the spinal cord area; transdermal; or via rectal suppository. It acts directly on the central nervous system (CNS) to induce analgesia and alter perception and emotional response to pain. Physical and psychological dependence and tolerance may develop with repeated administration. It can be taken for both acute pain and chronic pain and is frequently used for pain from myocardial infarction, kidney stones, and during labor. Its maximum effect is reached after about 20 minutes when administered intravenously and 60 minutes when administered by mouth, while the duration of its effect is 3–7 hours. Long-acting formulations of morphine are sold under the brand names MS Contin and Kadian, among others. Generic long-acting formulations are also available.

Hydromorphone, also known as dihydromorphinone, and sold under the brand name Dilaudid among others, is a morphinan opioid used to treat moderate to severe pain. Typically, long-term use is only recommended for pain due to cancer. It may be used by mouth or by injection into a vein, muscle, or under the skin. Effects generally begin within half an hour and last for up to five hours. A 2016 Cochrane review found little difference in benefit between hydromorphone and other opioids for cancer pain.

Opioids are a class of drugs that derive from, or mimic, natural substances found in the opium poppy plant. Opioids work in the brain to produce a variety of effects, including pain relief. As a class of substances, they act on opioid receptors to produce morphine-like effects.

Oxymorphone is a highly potent opioid analgesic indicated for treatment of severe pain. Pain relief after injection begins after about 5–10 minutes, after oral administration it begins after about 30 minutes, and lasts about 3–4 hours for immediate-release tablets and 12 hours for extended-release tablets. The elimination half-life of oxymorphone is much faster intravenously, and as such, the drug is most commonly used orally. Like oxycodone, which metabolizes to oxymorphone, oxymorphone has a high potential to be abused.

Codeinone is an isoquinolone alkaloid found in the opium poppy. As an analgesic, it is one-third the potency of codeine. It is an important intermediate in the production of hydrocodone–a painkiller about three-quarters the potency of morphine–as well as of oxycodone, though the latter can also be synthesized from thebaine.
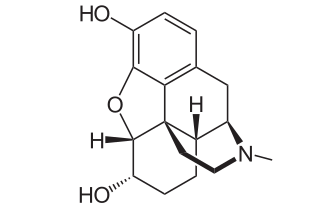
Dihydromorphine is a semi-synthetic opioid structurally related to and derived from morphine. The 7,8-double bond in morphine is reduced to a single bond to get dihydromorphine. Dihydromorphine is a moderately strong analgesic and is used clinically in the treatment of pain and also is an active metabolite of the analgesic opioid drug dihydrocodeine. Dihydromorphine occurs in trace quantities in assays of opium on occasion, as does dihydrocodeine, dihydrothebaine, tetrahydrothebaine, etc. The process for manufacturing dihydromorphine from morphine for pharmaceutical use was developed in Germany in the late 19th century, with the synthesis being published in 1900 and the drug introduced clinically as Paramorfan shortly thereafter. A high-yield synthesis from tetrahydrothebaine was later developed.

Levorphanol is an opioid medication used to treat moderate to severe pain. It is the levorotatory enantiomer of the compound racemorphan. Its dextrorotatory counterpart is dextrorphan.
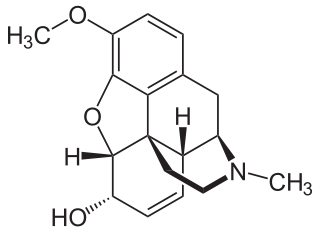
Codeine is an opiate and prodrug of morphine mainly used to treat pain, coughing, and diarrhea. It is also commonly used as a recreational drug. It is found naturally in the sap of the opium poppy, Papaver somniferum. It is typically used to treat mild to moderate degrees of pain. Greater benefit may occur when combined with paracetamol (acetaminophen) or a nonsteroidal anti-inflammatory drug (NSAID) such as aspirin or ibuprofen. Evidence does not support its use for acute cough suppression in children. In Europe, it is not recommended as a cough medicine in those under 12 years of age. It is generally taken by mouth. It typically starts working after half an hour, with maximum effect at two hours. Its effects last for about four to six hours. Codeine exhibits abuse potential similar to other opioid medications, including a risk of addiction and overdose.

Dihydroetorphine was developed by K. W. Bentley at McFarlan-Smith in the 1960s and is a potent opioid analgesic used mainly in China. It is a derivative of the better-known opioid etorphine, a very potent veterinary painkiller and anesthetic medication used primarily for the sedation of large animals such as elephants, giraffes, and rhinos.

Oripavine is an opioid and the major metabolite of thebaine. It is the parent compound from which a series of semi-synthetic opioids are derived, which includes the compounds etorphine and buprenorphine. Although its analgesic potency is comparable to morphine, it is not used clinically due to its severe toxicity and low therapeutic index. Being a precursor to a series of extremely strong opioids, oripavine is a controlled substance in some jurisdictions.
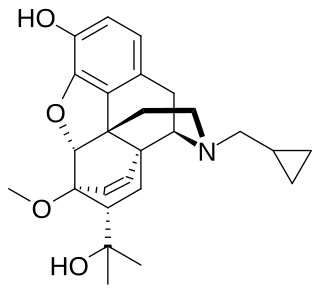
Cyprenorphine (M285), N-cyclo-propylmethyl-6,14-endoetheno-7α-(1-hydroxy-1-methylethyl)-6,7,8,14-tetrahydronororipavine, is an opioid drug. It is related to more well-known opioids such as buprenorphine, which is used as an analgesic and for the treatment of opioid addiction, and diprenorphine, which is used as an antidote to reverse the effects of other opioids. It is roughly 35 times as strong as nalorphine.

Meptazinol, sold under the brand name Meptid, is an opioid analgesic developed by Wyeth in the 1970s. Indications for use in moderate to severe pain, most commonly used to treat pain in obstetrics (childbirth).

Proglumide, sold under the brand name Milid, is a drug that inhibits gastrointestinal motility and reduces gastric secretions. It acts as a cholecystokinin antagonist, which blocks both the CCKA and CCKB subtypes. It was used mainly in the treatment of stomach ulcers, although it has now been largely replaced by newer drugs for this application.
DAMGO is a synthetic opioid peptide with high μ-opioid receptor specificity. It was synthesized as a biologically stable analog of δ-opioid receptor-preferring endogenous opioids, leu- and met-enkephalin. Structures of DAMGO bound to the μ opioid receptor reveal a very similar binding pose to morphinans.

Pentamorphone is a semi-synthetic opiate derivative related to compounds such as Morphinone and oxymorphone. Developed in 1984, it is a potent opioid analgesic several times stronger than fentanyl, and with a similarly fast onset of effects and short duration of action. It was found to produce relatively little respiratory depression compared to other potent opioid agonists, but its analgesic effects were somewhat disappointing in human trials, and while pentamorphone had some slight advantages over fentanyl these were not sufficient to warrant its introduction into clinical use.

An opiate is an alkaloid substance derived from opium. It differs from the similar term opioid in that the latter is used to designate all substances, both natural and synthetic, that bind to opioid receptors in the brain. Opiates are alkaloid compounds naturally found in the opium poppy plant Papaver somniferum. The psychoactive compounds found in the opium plant include morphine, codeine, and thebaine. Opiates have long been used for a variety of medical conditions, with evidence of opiate trade and use for pain relief as early as the eighth century AD. Most opiates are considered drugs with moderate to high abuse potential and are listed on various "Substance-Control Schedules" under the Uniform Controlled Substances Act of the United States of America.

PZM21 is an experimental opioid analgesic drug that is being researched for the treatment of pain. It is claimed to be a functionally selective μ-opioid receptor agonist which produces μ-opioid receptor mediated G protein signaling, with potency and efficacy similar to morphine, but with less β-arrestin 2 recruitment. However, recent reports highlight that this might be due to its low intrinsic efficacy, rather than functional selectivity or 'G protein bias' as initially reported. In tests on mice, PZM21 was slightly less potent than morphine or TRV130 as an analgesic, but also had significantly reduced adverse effects, with less constipation than morphine, and very little respiratory depression, even at high doses. This research was described as a compelling example of how modern high-throughput screening techniques can be used to discover new chemotypes with specific activity profiles, even at targets such as the μ-opioid receptor which have already been thoroughly investigated. More recent research has suggested however that at higher doses, PZM21 is capable of producing classic opioid side effects such as respiratory depression and development of tolerance and may have only limited functional selectivity.
Morphinone reductase is an enzyme which catalyzes the NADH-dependent saturation of the carbon-carbon double bond of morphinone and codeinone, yielding hydromorphone and hydrocodone respectively. This saturation reaction is assisted by a FMN cofactor and the enzyme is a member of the α/β-barrel flavoprotein family. The sequence of the enzyme has been obtained from bacteria Pseudomonas putida M10 and has been successfully expressed in yeast and other bacterial species. The enzyme is reported to harbor high sequence and structural similarity to the Old Yellow Enzyme, a large group of flavin-dependent redox biocatalysts of yeast species, and an oestrogen-binding protein of Candida albicans. The enzyme has demonstrated value in biosynthesis of semi-opiate drugs in microorganisms, expanding the chemical diversity of BIA biosynthesis.

















