| Moss | |
|---|---|
 | |
| Clumps of moss on the ground and base of trees in the Allegheny National Forest, Pennsylvania, United States | |
| Scientific classification | |
| Kingdom: | Plantae |
| Clade: | Embryophytes |
| Clade: | Setaphyta |
| Division: | Bryophyta Schimp. sensu stricto |
| Classes [2] | |
| Synonyms | |
| |
Mosses are small, non-vascular flowerless plants in the taxonomic division Bryophyta ( /braɪˈɒfətə/ , [3] /ˌbraɪ.əˈfaɪtə/ ) sensu stricto . Bryophyta ( sensu lato , Schimp. 1879 [4] ) may also refer to the parent group bryophytes, which comprise liverworts, mosses, and hornworts. [5] Mosses typically form dense green clumps or mats, often in damp or shady locations. The individual plants are usually composed of simple leaves that are generally only one cell thick, attached to a stem that may be branched or unbranched and has only a limited role in conducting water and nutrients. Although some species have conducting tissues, these are generally poorly developed and structurally different from similar tissue found in vascular plants. [6] Mosses do not have seeds and after fertilisation develop sporophytes with unbranched stalks topped with single capsules containing spores. They are typically 0.2–10 cm (0.1–3.9 in) tall, though some species are much larger. Dawsonia, the tallest moss in the world, can grow to 50 cm (20 in) in height. There are approximately 12,000 species. [2]
Contents
- Physical characteristics
- Description
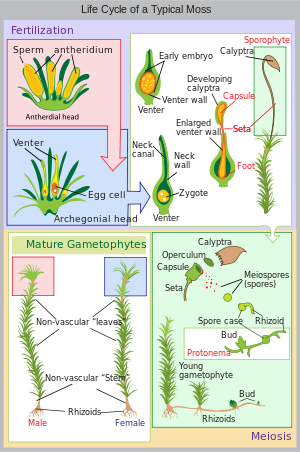
- Life cycle
- Dwarf males
- DNA repair
- Classification
- Geological history
- Ecology
- Habitat
- Relationship with cyanobacteria
- Cultivation
- Green roofs and walls
- Mossery
- Aquascaping
- Growth inhibition
- Uses
- Traditional
- Commercial
- References
- Further reading
- External links
Mosses are commonly confused with liverworts, hornworts and lichens. [7] Although often described as non-vascular plants, many mosses have advanced vascular systems. [8] [9] Like liverworts and hornworts, the haploid gametophyte generation of mosses is the dominant phase of the life cycle. This contrasts with the pattern in all vascular plants (seed plants and pteridophytes), where the diploid sporophyte generation is dominant. Lichens may superficially resemble mosses, and sometimes have common names that include the word "moss" (e.g., "reindeer moss" or "Iceland moss"), but they are fungal symbioses and not related to mosses. [7] : 3
The main commercial significance of mosses is as the main constituent of peat (mostly the genus Sphagnum ), although they are also used for decorative purposes, such as in gardens and in the florist trade. Traditional uses of mosses included as insulation and for the ability to absorb liquids up to 20 times their weight. Moss is a keystone genus and benefits habitat restoration and reforestation. [10]