
α-Methylfentanyl an opioid analgesic that is an analog of fentanyl. It is sometimes sold as "China White".

Ocfentanil is a potent synthetic opioid structurally related to fentanyl that was developed in the early 1990s as one of a series of potent naloxone-reversible opioids in an attempt to obtain an opioid that had better therapeutic indices in terms of cardiovascular effects and respiratory depression as compared to fentanyl. Ocfentanil was never developed for medical use despite reasonable results in human clinical trials, but subsequently started to be sold as a designer drug starting in around 2013.

Butyrfentanyl or butyrylfentanyl is a potent short-acting synthetic opioid analgesic drug. It is an analog of fentanyl with around one quarter of its potency. One of the first mentions of this drug can be found in document written by The College on Problem of Drug Dependence, where it is mentioned as N-butyramide fentanyl analog. This document also states that the article describing its clinical effects was published in 1987. It is an agonist for the μ-opioid receptors.

U-47700, also known as U4, pink heroin, pinky, and pink, is an opioid analgesic drug developed by a team at Upjohn in the 1970s which has around 7.5 times the potency of morphine in animal models.

Acetylfentanyl is an opioid analgesic drug that is an analog of fentanyl. Studies have estimated acetylfentanyl to be 15 times more potent than morphine, which would mean that despite being somewhat weaker than fentanyl, it is nevertheless still several times stronger than pure heroin. It has never been licensed for medical use and instead has only been sold as a designer drug. Acetylfentanyl was discovered at the same time as fentanyl itself and had only rarely been encountered on the illicit market in the late 1980s. However, in 2013, Canadian police seized 3 kilograms of acetylfentanyl. As a μ-opioid receptor agonist, acetylfentanyl may serve as a direct substitute for heroin or other opioids. Common side effects of fentanyl analogs are similar to those of fentanyl itself, which include itching, nausea and potentially serious respiratory depression, which can be life-threatening. Fentanyl analogs have killed hundreds of people throughout Europe and the former Soviet republics since the most recent resurgence in use began in Estonia in the early 2000s, and novel derivatives continue to appear.
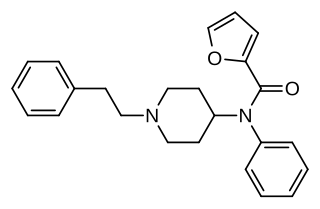
Furanylfentanyl (Fu-F) is an opioid analgesic that is an analog of fentanyl and has been sold as a designer drug. It has an ED50 value of 0.02 mg/kg in mice. This makes it approximately one fifth as potent as fentanyl.

3-Methylbutyrfentanyl (3-MBF) is an opioid analgesic that is an analog of butyrfentanyl.

Isofentanyl (3-methyl-benzylfentanyl) is an opioid analgesic that is an analog of fentanyl first invented in 1973, and which has been sold as a designer drug.

Butyrnorfentanyl or butyrylnorfentanyl is an inactive synthetic opioid analgesic drug precursor. It is an analog of fentanyl.

Despropionyl-p-fluorofentanyl is an inactive synthetic opioid analgesic drug precursor to 4-fluorofentanyl. It is an analog of fentanyl.

Furanylnorfentanyl is an inactive synthetic opioid analgesic drug precursor. It is an analog of fentanyl.

para-Methoxyfentanyl or p-methoxyfentanyl or 4-methoxyfentanyl is a potent short-acting synthetic opioid analgesic drug. It is an analog of fentanyl, with similar effects but slightly lower potency.

Remifentanilic acid is a metabolite of the potent short-acting synthetic opioid analgesic drug remifentanil. It is an analog of fentanyl and remifentanil, but is not active as an opioid in its own right.

Protonitazene is a benzimidazole derivative with potent opioid effects which has been sold over the internet as a designer drug since 2019, and has been identified in various European countries, as well as Canada, the USA and Australia. It has been linked to numerous cases of drug overdose, and is a Schedule I drug in the USA.

para-Fluorofuranylfentanyl is an opioid analgesic that is an analog of fentanyl and has been sold as a designer drug. As with other fentanyl analogues, parafluorofuranylfentanyl has significant side effects including itching, nausea and potentially serious respiratory depression, which can be life-threatening, and it has been linked to numerous deaths from overdose. It falls within the definition of Schedule I drugs in the USA under federal drug analogue legislation, and is specifically listed as a Schedule I drug in North Dakota.

N-Desethylisotonitazene (Norisotonitazene) is a benzimidazole derivative with potent opioid effects which has been sold as a designer drug. It was first identified as an active metabolite of the related compound isotonitazene, but was unexpectedly found to be similar potency compared to the parent compound, and is among the most potent opioid agonists in this family, around 20 times stronger than fentanyl. It has become an increasingly widespread drug of abuse in its own right, linked to numerous cases of drug overdose,and may be considered an analog of a schedule 1 drug in the USA. On October 25th an intent to temporarily schedule Etonitazepipne and N-desethyl Isotonitazene was published.So on November 24th a month after publishing intent, it will most likely be placed in schedule 1.

U-48800 is an opioid analgesic which has been sold as a designer drug. Unlike U-47700, it is primarily active as a kappa opioid receptor agonist with only moderate affinity at the mu opioid receptor. Nevertheless, it has still appeared on the recreational drug market, often as a component of combinations with other drugs, and has been linked to numerous drug overdose cases.

Flunitazene (Fluonitazene) is a benzimidazole derivative with opioid effects, first developed in the 1950s as part of the research that led to better-known compounds such as etonitazene. It is one of the least potent derivatives from this class to have appeared as a designer drug, with only around the same potency as morphine, but nevertheless has been sold since around 2020, and has been linked to numerous drug overdose cases.



















