 | |
 | |
 | |
| Names | |
|---|---|
| IUPAC name Nitrosyl chloride [1] | |
| Identifiers | |
3D model (JSmol) | |
| ChemSpider | |
| ECHA InfoCard | 100.018.430 |
| EC Number |
|
| E number | E919 (glazing agents, ...) |
| MeSH | nitrosyl+chloride |
PubChem CID | |
| RTECS number |
|
| UNII | |
| UN number | 1069 |
CompTox Dashboard (EPA) | |
| |
| |
| Properties | |
| NOCl | |
| Molar mass | 65.459 g mol−1 |
| Appearance | yellow gas |
| Density | 2.872 mg mL−1 |
| Melting point | −59.4 °C (−74.9 °F; 213.8 K) |
| Boiling point | −5.55 °C (22.01 °F; 267.60 K) |
| Reacts | |
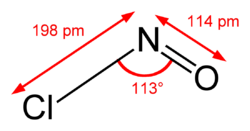
| Structure | |
| Dihedral, digonal | |
| Hybridisation | sp2 at N |
| 1.90 D | |
| Thermochemistry | |
Std molar entropy (S⦵298) | 261.68 J K−1 mol−1 |
Std enthalpy of formation (ΔfH⦵298) | 51.71 kJ mol−1 |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Safety data sheet (SDS) | inchem.org |
| Related compounds | |
Related compounds | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
Nitrosyl chloride is the chemical compound with the formula NOCl. It is a yellow gas that is commonly encountered as a component of aqua regia, a mixture of 3 parts concentrated hydrochloric acid and 1 part of concentrated nitric acid. It is a strong electrophile and oxidizing agent. It is sometimes called Tilden's reagent, after William A. Tilden, who was the first to produce it as a pure compound. [2]

