Weasels are mammals of the genus Mustela of the family Mustelidae. The genus Mustela includes the least weasels, polecats, stoats, ferrets, and European mink. Members of this genus are small, active predators, with long and slender bodies and short legs. The family Mustelidae, or mustelids, is often referred to as the "weasel family". In the UK, the term "weasel" usually refers to the smallest species, the least weasel (M. nivalis), the smallest carnivoran species.

The weasel war dance is a colloquial term for the behavior of excited ferrets and other members of the weasel subfamily. Naturalists speculate that weasels in the wild use the dance to confuse or disorient prey.

The wildcat is a species complex comprising two small wild cat species: the European wildcat and the African wildcat. The European wildcat inhabits forests in Europe, Anatolia and the Caucasus, while the African wildcat inhabits semi-arid landscapes and steppes in Africa, the Arabian Peninsula, Central Asia, into western India and western China. The wildcat species differ in fur pattern, tail, and size: the European wildcat has long fur and a bushy tail with a rounded tip; the smaller African wildcat is more faintly striped, has short sandy-gray fur and a tapering tail; the Asiatic wildcat is spotted.

The European polecat, also known as the common polecat, black polecat and forest polecat, is a mustelid species native to western Eurasia and North Africa. It is of a generally dark brown colour, with a pale underbelly and a dark mask across the face. Occasionally, colour mutations including albinos, leucists, isabellinists, xanthochromists, amelanists and erythrists occur. It has a shorter, more compact body than other Mustela species, a more powerfully built skull and dentition, is less agile, and is well known for having the characteristic ability to secrete a particularly foul-smelling liquid to mark its territory.

The honey badger, also known as the ratel, is a mammal widely distributed in Africa, Southwest Asia, and the Indian subcontinent. Because of its wide range and occurrence in a variety of habitats, it is listed as Least Concern on the IUCN Red List.

The European badger, also known as the Eurasian badger, is a badger species in the family Mustelidae native to Europe and West Asia and parts of Central Asia. It is classified as least concern on the IUCN Red List, as it has a wide range and a large, stable population size which is thought to be increasing in some regions. Several subspecies are recognized, with the nominate subspecies predominating in most of Europe. In Europe, where no other badger species commonly occurs, it is generally just called the "badger".
| name = Long-tailed weasel | image = Mustela_frenata_new.jpg | status = LC | status_system = IUCN3.1 | status_ref = | genus = Neogale | species = frenata | authority = | range_map = Long-tailed Weasel area.png | range_map_caption = Long-tailed weasel range | synonyms = Mustela frenata }}

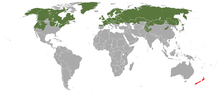
The least weasel, little weasel, common weasel, or simply weasel is the smallest member of the genus Mustela, family Mustelidae and order Carnivora. It is native to Eurasia, North America and North Africa, and has been introduced to New Zealand, Malta, Crete, the Azores, and São Tomé. It is classified as least concern by the IUCN, due to its wide distribution and large population throughout the Northern Hemisphere.

The European mink, also known as the Russian mink and Eurasian mink, is a semiaquatic species of mustelid native to Europe.

The American mink is a semiaquatic species of mustelid native to North America, though human introduction has expanded its range to many parts of Europe, Asia, and South America. Because of range expansion, the American mink is classed as a least-concern species by the IUCN. The American mink was formerly thought to be the only extant member of the genus Neovison following the extinction of the sea mink (N. macrodon), but recent studies, followed by taxonomic authorities, have reclassified it and the sea mink within the genus Neogale, which also contains a few New World weasel species. The American mink is a carnivore that feeds on rodents, fish, crustaceans, frogs, and birds. In its introduced range in Europe it has been classified as an invasive species linked to declines in European mink, Pyrenean desman, and water vole populations. It is the animal most frequently farmed for its fur, exceeding the silver fox, sable, marten, and skunk in economic importance.

The steppe polecat, also known as the white or masked polecat, is a species of mustelid native to Central and Eastern Europe and Central and East Asia.It is listed as Least Concern on the IUCN Red List because of its wide distribution, occurrence in a number of protected areas, and tolerance to some degree of habitat modification. It is generally of a very light yellowish colour, with dark limbs and a dark mask across the face. Compared to its relative, the European polecat, the steppe polecat is larger in size and has a more powerfully built skull.

The beech marten, also known as the stone marten, house marten or white breasted marten, is a species of marten native to much of Europe and Central Asia, though it has established a feral population in North America. It is listed as Least Concern on the IUCN Red List on account of its wide distribution, its large population, and its presence in a number of protected areas. It is superficially similar to the European pine marten, but differs from it by its smaller size and habitat preferences. While the pine marten is a forest specialist, the beech marten is a more generalist and adaptable species, occurring in a number of open and forest habitats.

The Siberian weasel or kolonok, is a medium-sized weasel native to Asia, where it is widely distributed and inhabits various forest habitats and open areas. It is therefore listed as Least Concern on the IUCN Red List.

The mountain weasel, also known as the pale weasel, Altai weasel or solongoi, primarily lives in high-altitude environments, as well as rocky tundra and grassy woodlands. This weasel rests in rock crevices, tree trunks, and abandoned burrows of other animals or the animals it previously hunted. The home range size of this animal is currently unknown. Geographical distribution for this species lies in parts of Asia from Kazakhstan, Tibet, and the Himalayas to Mongolia, northeastern China, and southern Siberia. The most common area for this species, however, is Ladakh, India. The conservation status, according to the IUCN, is near threatened because it is considered to be in significant decline and requires monitoring mainly because of habitat and resource loss.

The yellow-throated marten is a marten species native to the Himalayas, Southeast and East Asia. Its coat is bright yellow-golden, and its head and back are distinctly darker, blending together black, white, golden-yellow and brown. It is the second-largest marten in the Old World, after the Nilgiri marten, with its tail making up more than half its body length.

The Colombian weasel, also known as Don Felipe's weasel, is a very rare species of weasel only known with certainty from the departments of Huila and Cauca in Colombia and nearby northern Ecuador. Both its scientific and alternative common name honours the mammalogist Philip "Don Felipe" Hershkovitz.

The Japanese weasel is a carnivorous mammal belonging to the genus Mustela in the family Mustelidae. The most closely related Mustela species is the Siberian weasel. Its taxonomic species name, itatsi, is based on the Japanese word for weasel, itachi (イタチ). It is native to Japan where it occurs on the islands of Honshū, Kyūshū and Shikoku. It has been introduced to Hokkaidō and the Ryukyu Islands to control rodents and has also been introduced to Sakhalin Island in Russia.

The stoat was introduced into New Zealand to control introduced rabbits and hares, but is now a major threat to the native bird population. The natural range of the stoat is limited to parts of the Northern Hemisphere. Immediately before human settlement, New Zealand did not have any land-based mammals apart from bats, but Polynesian and European settlers introduced a wide variety of animals. Rarely, in Southland, the fur of stoats has been reported to turn white, being the fur known as ermine, which adorns royal robes.

The Haida ermine is a mustelid species endemic to a few islands off the Pacific Northwest of North America, namely Haida Gwaii in Canada and the southern Alexander Archipelago in the U.S. state of Alaska.

The American ermine or American stoat is a species of mustelid native to most of North America.