
Salvinorin A is the main active psychotropic molecule in Salvia divinorum. Salvinorin A is considered a dissociative hallucinogen.

Piperazine is an organic compound that consists of a six-membered ring containing two nitrogen atoms at opposite positions in the ring. Piperazine exists as small alkaline deliquescent crystals with a saline taste.

Benzothiazole, or more specifically 1,3-benzothiazole, is an aromatic heterocyclic compound with the chemical formula C
7H
5NS. It is colorless, slightly viscous liquid. Although the parent compound, benzothiazole is not widely used, many of its derivatives are found in commercial products or in nature. Firefly luciferin can be considered a derivative of benzothiazole.

Dextromoramide is a powerful opioid analgesic approximately three times more potent than morphine but shorter acting. It is subject to drug prohibition regimes, both internationally through UN treaties and by the criminal law of individual nations, and is usually prescribed only in the Netherlands.

Etonitazene, also known as EA-4941 or CS-4640, is a benzimidazole opioid, first reported in 1957, that has been shown to have approximately 1,000 to 1,500 times the potency of morphine in animals.

Lefetamine (Santenol) is a drug which is a stimulant and also an analgesic with effects comparable to codeine.

Tandospirone, sold under the brand name Sediel, is an anxiolytic and antidepressant medication used in Japan and China, where it is marketed by Dainippon Sumitomo Pharma. It is a member of the azapirone class of drugs and is closely related to other azapirones like buspirone and gepirone.

Azaprocin is a drug which is an opioid analgesic with approximately ten times the potency of morphine, and a fast onset and short duration of action. It was discovered in 1963, but has never been marketed.

Substituted phenethylamines are a chemical class of organic compounds that are based upon the phenethylamine structure; the class is composed of all the derivative compounds of phenethylamine which can be formed by replacing, or substituting, one or more hydrogen atoms in the phenethylamine core structure with substituents.

Arylcyclohexylamines, also known as arylcyclohexamines or arylcyclohexanamines, are a chemical class of pharmaceutical, designer, and experimental drugs.
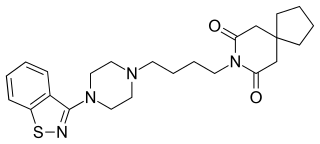
Tiospirone (BMY-13,859), also sometimes called tiaspirone or tiosperone, is an atypical antipsychotic of the azapirone class. It was investigated as a treatment for schizophrenia in the late 1980s and was found to have an effectiveness equivalent to those of typical antipsychotics in clinical trials but without causing extrapyramidal side effects. However, development was halted and it was not marketed. Perospirone, another azapirone derivative with antipsychotic properties, was synthesized and assayed several years after tiospirone. It was found to be both more potent and more selective in comparison and was commercialized instead.

Cannabicyclohexanol is a cannabinoid receptor agonist drug, developed by Pfizer in 1979. On 19 January 2009, the University of Freiburg in Germany announced that an analog of CP 47,497 was the main active ingredient in the herbal incense product Spice, specifically the 1,1-dimethyloctyl homologue of CP 47,497, which is now known as cannabicyclohexanol. The 1,1-dimethyloctyl homologue of CP 47,497 is in fact several times more potent than the parent compound, which is somewhat unexpected as the 1,1-dimethylheptyl is the most potent substituent in classical cannabinoid compounds such as HU-210.

MT-45 (IC-6) is an opioid analgesic drug invented in the 1970s by Dainippon Pharmaceutical Co. It is chemically a 1-substituted-4-(1,2-diphenylethyl) piperazine derivative, which is structurally unrelated to most other opioid drugs. Racemic MT-45 has around 80% the potency of morphine, with almost all opioid activity residing in the (S) enantiomer. It has been used as a lead compound from which a large family of potent opioid drugs have been developed, including full agonists, partial agonists, and antagonists at the three main opioid receptor subtypes. Fluorinated derivatives of MT-45 such as 2F-MT-45 are significantly more potent as μ-opioid receptor agonists, and one of its main metabolites 1,2-diphenylethylpiperazine also blocks NMDA receptors.

AD-1211 is an opioid analgesic drug invented in the 1970s by Dainippon Pharmaceutical Co. It is chemically a 1-substituted-4-prenyl-piperazine derivative, which is structurally unrelated to most other opioid drugs. The (S)-enantiomers in this series are more active as opioid agonists, but the less active (R)-enantiomer of this compound, AD-1211, is a mixed agonist–antagonist at opioid receptors with a similar pharmacological profile to pentazocine, and has atypical opioid effects with little development of tolerance or dependence seen after extended administration in animal studies.
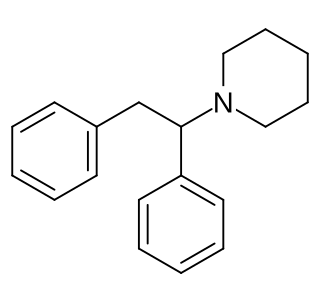
Diphenidine is a dissociative anesthetic that has been sold as a designer drug. The synthesis of diphenidine was first reported in 1924, and employed a Bruylants reaction analogous to the one that would later be used to discover phencyclidine in 1956. Shortly after the 2013 UK ban on arylcyclohexylamines, diphenidine and the related compound methoxphenidine became available on the grey market. Anecdotal reports describe high doses of diphenidine producing "bizarre somatosensory phenomena and transient anterograde amnesia." Diphenidine and related diarylethylamines have been studied in vitro as treatments for neurotoxic injury and are antagonists of the NMDA receptor. In dogs diphenidine exhibits greater antitussive potency than codeine phosphate.

Ephenidine is a dissociative anesthetic that has been sold online as a designer drug. It is illegal in some countries as a structural isomer of the banned opioid drug lefetamine, but has been sold in countries where it is not yet banned.
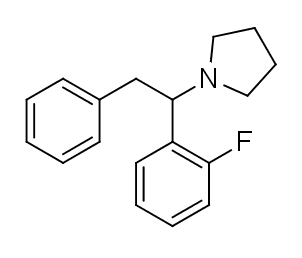
Fluorolintane is a dissociative anesthetic drug that has been sold online as a designer drug.
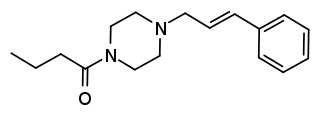
Bucinnazine is an opioid analgesic drug that was widely used in China to treat pain in cancer patients as of 1986. It is one of the most potent compounds among a series of piperazine-amides first synthesized and reported in Japan in the 1970s. Bucinnazine has analgesic potency comparable to that of morphine but with a relatively higher therapeutic index.

1,2-Dioxolane is a chemical compound with formula C3H6O2, consisting of a ring of three carbon atoms and two oxygen atoms in adjacent positions. Its condensed structural formula is [–(CH
2)3–O–O–].




















