A local anesthetic (LA) is a medication that causes absence of all sensation in a specific body part without loss of consciousness, providing local anesthesia, as opposed to a general anesthetic, which eliminates all sensation in the entire body and causes unconsciousness. Local anesthetics are most commonly used to eliminate pain during or after surgery. When it is used on specific nerve pathways, paralysis also can be induced.

Cold medicines are a group of medications taken individually or in combination as a treatment for the symptoms of the common cold and similar conditions of the upper respiratory tract. The term encompasses a broad array of drugs, including analgesics, antihistamines and decongestants, among many others. It also includes drugs which are marketed as cough suppressants or antitussives, but their effectiveness in reducing cough symptoms is unclear or minimal.

Procaine is a local anesthetic drug of the amino ester group. It is most commonly used in dental procedures to numb the area around a tooth and is also used to reduce the pain of intramuscular injection of penicillin. Owing to the ubiquity of the trade name Novocain or Novocaine, in some regions, procaine is referred to generically as novocaine. It acts mainly as a sodium channel blocker. Today, it is used therapeutically in some countries due to its sympatholytic, anti-inflammatory, perfusion-enhancing, and mood-enhancing effects.

Lidocaine, also known as lignocaine and sold under the brand name Xylocaine among others, is a local anesthetic of the amino amide type. It is also used to treat ventricular tachycardia and ventricular fibrillation. When used for local anaesthesia or in nerve blocks, lidocaine typically begins working within several minutes and lasts for half an hour to three hours. Lidocaine mixtures may also be applied directly to the skin or mucous membranes to numb the area. It is often used mixed with a small amount of adrenaline (epinephrine) to prolong its local effects and to decrease bleeding.

Benadryl is a brand of various antihistamine medications used to stop allergies, whose content varies in different countries, but which includes some combination of diphenhydramine, acrivastine, and/or cetirizine.

An anesthetic or anaesthetic is a drug used to induce anesthesia — in other words, to result in a temporary loss of sensation or awareness. They may be divided into two broad classes: general anesthetics, which result in a reversible loss of consciousness, and local anesthetics, which cause a reversible loss of sensation for a limited region of the body without necessarily affecting consciousness.

Buprenorphine, sold under the brand name Subutex among others, is an opioid used to treat opioid use disorder, acute pain, and chronic pain. It can be used under the tongue (sublingual), in the cheek (buccal), by injection, as a skin patch (transdermal), or as an implant. For opioid use disorder, the patient must have moderate opioid withdrawal symptoms before buprenorphine can be administered under direct observation of a health-care provider.
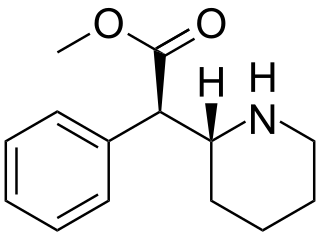
Dexmethylphenidate, sold under the brand name Focalin among others, is a potent central nervous system (CNS) stimulant used to treat attention deficit hyperactivity disorder (ADHD) in those over the age of five years. It is taken by mouth. The immediate release formulation lasts up to five hours while the extended release formulation lasts up to twelve hours. It is the more active enantiomer of methylphenidate.
Vicks DayQuil is an over-the-counter combination medication product used for the temporary relief of common cold and flu symptoms. DayQuil is available in several formulations.

Guaifenesin, also known as glyceryl guaiacolate, is an expectorant medication taken by mouth and marketed as an aid to eliminate sputum from the respiratory tract. Chemically, it is an ether of guaiacol and glycerine. It may be used in combination with other medications. A 2014 study found that guaifenesin has no effect on sputum volume in upper respiratory infections.

Dextropropoxyphene is an analgesic in the opioid category, patented in 1955 and manufactured by Eli Lilly and Company. It is an optical isomer of levopropoxyphene. It is intended to treat mild pain and also has antitussive and local anaesthetic effects. The drug has been taken off the market in Europe and the US due to concerns of fatal overdoses and heart arrhythmias. It is still available in Australia, albeit with restrictions after an application by its manufacturer to review its proposed banning. Its onset of analgesia is said to be 20–30 minutes and peak effects are seen about 1.5–2.0 hours after oral administration.

Betaxolol is a selective beta1 receptor blocker used in the treatment of hypertension and angina. It is also a adrenergic blocker with no partial agonist action and minimal membrane stabilizing activity. Being selective for beta1 receptors, it typically has fewer systemic side effects than non-selective beta-blockers, for example, not causing bronchospasm as timolol may. Betaxolol also shows greater affinity for beta1 receptors than metoprolol. In addition to its effect on the heart, betaxolol reduces the pressure within the eye. This effect is thought to be caused by reducing the production of the liquid within the eye. The precise mechanism of this effect is not known. The reduction in intraocular pressure reduces the risk of damage to the optic nerve and loss of vision in patients with elevated intraocular pressure due to glaucoma.

Levothyroxine, also known as L-thyroxine, is a synthetic form of the thyroid hormone thyroxine (T4). It is used to treat thyroid hormone deficiency (hypothyroidism), including a severe form known as myxedema coma. It may also be used to treat and prevent certain types of thyroid tumors. It is not indicated for weight loss. Levothyroxine is taken orally (by mouth) or given by intravenous injection. Levothyroxine has a half-life of 7.5 days when taken daily, so about six weeks is required for it to reach a steady level in the blood.

Bupivacaine, marketed under the brand name Marcaine among others, is a medication used to decrease sensation in a specific small area. In nerve blocks, it is injected around a nerve that supplies the area, or into the spinal canal's epidural space. It is available mixed with a small amount of epinephrine to increase the duration of its action. It typically begins working within 15 minutes and lasts for 2 to 8 hours.

Dextromethorphan (DXM), sold under the trade name Robitussin among others, is a cough suppressant used in many cough and cold medicines. In 2022, the US Food and Drug Administration (FDA) approved the combination dextromethorphan/bupropion to serve as a rapid-acting antidepressant in patients with major depressive disorder.
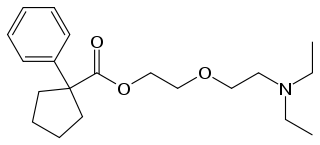
Pentoxyverine (rINN) or carbetapentane is an antitussive commonly used for cough associated with illnesses like common cold. It is sold over-the-counter as Solotuss, or in combination with other medications, especially decongestants. One such product is Certuss, a combination of guaifenesin and pentoxyverine. The drug has been available in the form of drops, suspensions and suppositories.
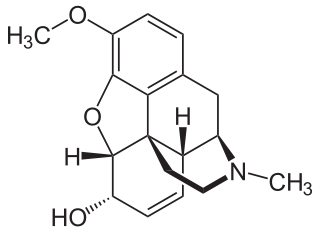
Codeine is an opiate and prodrug of morphine mainly used to treat pain, coughing, and diarrhea. It is also commonly used as a recreational drug. It is found naturally in the sap of the opium poppy, Papaver somniferum. It is typically used to treat mild to moderate degrees of pain. Greater benefit may occur when combined with paracetamol (acetaminophen) or a nonsteroidal anti-inflammatory drug (NSAID) such as aspirin or ibuprofen. Evidence does not support its use for acute cough suppression in children. In Europe, it is not recommended as a cough medicine for those under 12 years of age. It is generally taken by mouth. It typically starts working after half an hour, with maximum effect at two hours. Its effects last for about four to six hours. Codeine exhibits abuse potential similar to other opioid medications, including a risk of addiction and overdose.

An opioid overdose is toxicity due to excessive consumption of opioids, such as morphine, codeine, heroin, fentanyl, tramadol, and methadone. This preventable pathology can be fatal if it leads to respiratory depression, a lethal condition that can cause hypoxia from slow and shallow breathing. Other symptoms include small pupils and unconsciousness; however, its onset can depend on the method of ingestion, the dosage and individual risk factors. Although there were over 110,000 deaths in 2017 due to opioids, individuals who survived also faced adverse complications, including permanent brain damage.

Benzodiazepine overdose describes the ingestion of one of the drugs in the benzodiazepine class in quantities greater than are recommended or generally practiced. The most common symptoms of overdose include central nervous system (CNS) depression, impaired balance, ataxia, and slurred speech. Severe symptoms include coma and respiratory depression. Supportive care is the mainstay of treatment of benzodiazepine overdose. There is an antidote, flumazenil, but its use is controversial.

A psychoactive drug, mind-altering drug, or consciousness-altering drug is a chemical substance that changes brain function and results in alterations in perception, mood, consciousness, cognition, or behavior. The term psychotropic drug is often used interchangeably, while some sources present narrower definitions. These substances may be used medically; recreationally; to purposefully improve performance or alter consciousness; as entheogens for ritual, spiritual, or shamanic purposes; or for research, including psychedelic therapy. Physicians and other healthcare practitioners prescribe psychoactive drugs from several categories for therapeutic purposes. These include anesthetics, analgesics, anticonvulsant and antiparkinsonian drugs as well as medications used to treat neuropsychiatric disorders, such as antidepressants, anxiolytics, antipsychotics, and stimulants. Some psychoactive substances may be used in detoxification and rehabilitation programs for persons dependent on or addicted to other psychoactive drugs.