Structure
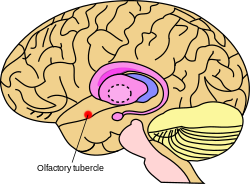
The olfactory tubercle differs in location and relative size between humans, other primates, rodents, birds, and other animals. In most cases, the olfactory tubercle is identified as a round bulge along the basal forebrain anterior to the optic chiasm and posterior to the olfactory peduncle. [7] In humans and other primates, visual identification of the olfactory tubercle is not easy because the basal forebrain bulge is small in these animals. [8] With regard to functional anatomy, the olfactory tubercle can be considered to be a part of three larger networks. First, it is considered to be part of the basal forebrain, the nucleus accumbens, and the amygdaloid nuclei because of its location along the rostral ventral region of the brain, that is, the front-bottom part. Second, it is considered to be part of the olfactory cortex because it receives direct input from the olfactory bulb. Third, it is also considered to be part of the ventral striatum based on anatomy, neurochemical, and embryology data.
One of the most striking features of the olfactory tubercle is the closely packed crescent-shape cell clusters, which are located mostly in layer III and sometimes in layer II. These cell clusters, called the islands of Calleja, are innervated by dopaminergic projections from the nucleus accumbens and the substantia nigra, suggesting the role that the olfactory tubercle plays in the reward system.
The olfactory tubercle is a multi-sensory processing center due to the number of innervations going to and from other brain regions such as the amygdala, thalamus, hypothalamus, hippocampus, brain stem, auditory and visual sensory fibers, and a number of structures in the reward–arousal system, as well as the olfactory cortex. Due to its many innervations from other brain regions, the olfactory tubercle is involved in merging information across the senses, such as olfactory/audition and olfactory/visual integrations, possibly in a behaviorally relevant manner. Thus, damage to the olfactory tubercle is likely to affect the functionality of all these areas of the brain. Examples of such disruption include changes in normal odor-guided behavior, and impairments in modulating state and motivational behavior, [3] which are common in psychiatric disorders such as schizophrenia, [9] dementia [10] and depression. [11]
The olfactory tubercle has been shown to play a large role in behavior. Unilateral lesions in the olfactory tubercle have been shown to alter attention, social and sensory responsiveness, and even locomotor behavior. [2] Bilateral lesions have been shown to reduce copulatory behavior in male rats. The olfactory tubercle has also been shown to be especially involved in reward and addictive behaviors. Rats have been shown to administer cocaine into the olfactory tubercle more than the nucleus accumbens and ventral pallidum, other reward centers in the brain. [12] In fact, they will administer cocaine into the olfactory tubercle at about 200 times per hour and even till death.
Functional contributions of the olfactory tubercle to olfaction are currently unclear; however, there is evidence of a perceptual role that it may play. Work from Zelano, et al. suggest that the olfactory tubercle may be crucial in sorting out the sources of olfactory information. [13] This suggests that it may also play a role in odor guided behavior. Thus, it may link perception of odor with action through its connections with attention, reward, and motivation systems of the basal forebrain. [3] Functional imaging data from this same group also shows that the olfactory tubercle is highly activated during tasks that engage attention, thus playing a large role in arousal-related systems.
Because the olfactory tubercle is a component of the ventral striatum, it is heavily interconnected with several affective-, reward-, and motivation-related centers of the brain. It also sits at the interface between the olfactory sensory input and state-dependent behavioral modulatory circuits, that is the area that modulates behavior during certain physiological and mental states. Thus, the olfactory tubercle may also play an important role in the mediation of odor approach and odor avoidance behavior, probably in a state-dependent manner. [14]
Anatomy
In general, the olfactory tubercle is located at the basal forebrain of the animal within the medial temporal lobe. Specifically, parts of the tubercle are included in the olfactory cortex and nested between the optic chiasm and olfactory tract and ventral to the nucleus accumbens. The olfactory tubercle consists of three layers, a molecular layer (layer I), the dense cell layer (layer II), and the multiform layer (layer III). [8] Other than the islands of Calleja, which are characteristic of the tubercle, [15] it is also noted for the being innervated by dopaminergic neurons from the ventral tegmental area. The olfactory tubercle also consists of heterogeneous elements, such as medial forebrain bundle, and has a ventral extension of the striatal complex. During the 1970s, the tubercle was found to contain a striatal component which is composed of GABAergic medium spiny neurons. [5] The GABAergic neurons project to the ventral pallidum and receive glutamatergic inputs from cortical regions and dopaminergic inputs from the ventral tegmental area. [16] [17]
Morphological and neurochemical features
The ventral portion of the olfactory tubercle consists of three layers, whereas the dorsal portion contains dense cell clusters and adjoins the ventral pallidum (within the basal ganglia). The structure of the most ventral and anterior parts of the tubercle can be defined as anatomically defined hills (consisting of gyri and sulci) and clusters of cells.
The most common cell types in the olfactory tubercle are medium-size dense spine cells found predominantly in layer II (dense cell layer). The dendrites of these cells are covered by substance p immunoreactive (S.P.I) axons up into layer III (multiform layer). [8] These cells also project into the nucleus accumbens and caudate putamen, thus linking the olfactory tubercle with the pallidum. [18] Other medium-size cells reside in layers II and III of the olfactory tubercle as well. These include the spine-poor neurons and spindle cells and they differ from the medium-size dense spine cells because they have sparse dendritic trees. The largest cells, and most striking feature of the olfactory tubercle, are densely packed crescent-shape cell clusters, Islands of Calleja that reside mostly in the dorsal portion of the olfactory tubercle, layer III, and can also be found in layer II. The olfactory tubercle also contains three classes of small cells found mostly in layers I and II. The first are pial cells (named as such because of location near pial surface), which look like miniature medium-size dense spine cells. The second are radiate cells and are easily identified by numerous multi-directional spineless dendrites. The third, small spine cells, are similar to the pial cells in that they also look like medium-size spine cells except they are not located near the pial surface. [19]
Development
Migrating cells from several developmental sites come together to form the olfactory tubercle. This includes the ventral ganglionic eminence (found in ventral part of telencephalon, where they form bulges in the ventricles that later become the basal ganglia, present only in embryonic stages) and the rostromedial telencephalic wall (of the forebrain). [20] Olfactory tubercle neurons originate as early as embryonic day 13 (E13), and the cell development occurs in a layer specific manner. The emergence of the three main layers of the olfactory tubercle begins almost simultaneously. The large neurons in layer III originate from E13 to E16, while the small and medium originate between E15 and E20. Like the small and medium cells in layer III, the cells of layer II and the striatal bridges also originate between E15 and E20 and develop in a lateral to medial gradient. [21] The granule cells of the islands of calleja originate between E19 and E22 and continue to migrate into the islands until long after birth. [22] [23]
Fibers from the lateral olfactory tract begin branching into the olfactory tubercle around E17. The lateral portion of the olfactory tubercle (which adjoins the olfactory tract) receives the densest fiber input and the medial portion receives light fiber projections. [24] This branching continues until completion about the end of the first week after birth.