
This is a list of selective estrogen receptor modulators (SERMs).

This is a list of selective estrogen receptor modulators (SERMs).
SERMs that have been approved for medical use include anordrin (+mifepristone (Zi Yun)), bazedoxifene (+conjugated estrogens (Duavee)), broparestrol (Acnestrol), clomifene (Clomid), cyclofenil (Sexovid), lasofoxifene (Fablyn), ormeloxifene (Centron, Novex, Novex-DS, Sevista), ospemifene (Osphena; deaminohydroxytoremifene), raloxifene (Evista), tamoxifen (Nolvadex), and toremifene (Fareston; 4-chlorotamoxifen). [1]
SERMs that are currently under development and in clinical trials include acolbifene, afimoxifene (4-hydroxytamoxifen; metabolite of tamoxifen), elacestrant, enclomifene ((E)-clomifene), endoxifen (4-hydroxy-N-desmethyltamoxifen; metabolite of tamoxifen), and zuclomifene ((Z)-clomifene). [2]
SERMs that have not been approved for medical use include arzoxifene, brilanestrant, clomifenoxide (clomiphene N-oxide; metabolite of clomifene), [3] droloxifene (3-hydroxytamoxifen), etacstil, fispemifene, GW-7604 (4-hydroxyetacstil; metabolite of etacstil), idoxifene (pyrrolidino-4-iodotamoxifen), levormeloxifene ((L)-ormeloxifene), miproxifene, nafoxidine, nitromifene (CI-628), NNC 45-0095, panomifene, pipendoxifene (ERA-923), trioxifene, and zindoxifene (D-16726). [4] [1] [5] [6] [7]
Sivifene (A-007) was initially thought to be a SERM due to its structural similarity to tamoxifen but it was subsequently found not to bind to the estrogen receptor (ER). [8] Tesmilifene (DPPE; YMB-1002, BMS-217380-01) is also structurally related to tamoxifen but similarly does not bind to the ER and is not a SERM. [9] [10]
SERMs can be variously classified structurally as triphenylethylenes (tamoxifen, clomifene, toremifene, droloxifene, idoxifene, ospemifene, fispemifene, afimoxifene, others), benzothiophenes (raloxifene, arzoxifene), indoles (bazedoxifene, zindoxifene, pipendoxifene), tetrahydronaphthalenes (lasofoxifene, nafoxidine), and benzopyrans (acolbifene, ormeloxifene, levormeloxifene). [11] [12] [13]

Clomifene, also known as clomiphene, is a medication used to treat infertility in women who do not ovulate, including those with polycystic ovary syndrome. Use results in a greater chance of twins. It is taken by mouth once a day, with a course of treatment that usually lasts for five days.

Selective estrogen receptor modulators (SERMs), also known as estrogen receptor agonist/antagonists (ERAAs), are a class of drugs that act on the estrogen receptor (ER). A characteristic that distinguishes these substances from pure ER agonists and antagonists is that their action is different in various tissues, thereby granting the possibility to selectively inhibit or stimulate estrogen-like action in various tissues.

Tamoxifen, sold under the brand name Nolvadex among others, is a selective estrogen receptor modulator used to prevent breast cancer in women and men. It is also being studied for other types of cancer. It has been used for Albright syndrome. Tamoxifen is typically taken daily by mouth for five years for breast cancer.

Bazedoxifene, used as bazedoxifene acetate, is a medication for bone problems and possibly for cancer. It is a third-generation selective estrogen receptor modulator (SERM). Since late 2013 it has had U.S. FDA approval for bazedoxifene as part of the combination drug Duavee in the prevention of postmenopausal osteoporosis. It is also being studied for possible treatment of breast cancer and pancreatic cancer.

Toremifene, sold under the brand name Fareston among others, is a medication which is used in the treatment of advanced breast cancer in postmenopausal women. It is taken by mouth.
A nonsteroidal compound is a drug that is not a steroid nor a steroid derivative. Nonsteroidal anti-inflammatory drugs (NSAIDs) are distinguished from corticosteroids as a class of anti-inflammatory agents.

Chlorotrianisene (CTA), also known as tri-p-anisylchloroethylene (TACE) and sold under the brand name Tace among others, is a nonsteroidal estrogen related to diethylstilbestrol (DES) which was previously used in the treatment of menopausal symptoms and estrogen deficiency in women and prostate cancer in men, among other indications, but has since been discontinued and is now no longer available. It is taken by mouth.

Lasofoxifene, sold under the brand name Fablyn, is a nonsteroidal selective estrogen receptor modulator (SERM) which is marketed by Pfizer in Lithuania and Portugal for the prevention and treatment of osteoporosis and for the treatment of vaginal atrophy, and the result of an exclusive research collaboration with Ligand Pharmaceuticals (LGND). It also appears to have had a statistically significant effect of reducing breast cancer in women according to a study published in The Journal of the National Cancer Institute.
Antiestrogens, also known as estrogen antagonists or estrogen blockers, are a class of drugs which prevent estrogens like estradiol from mediating their biological effects in the body. They act by blocking the estrogen receptor (ER) and/or inhibiting or suppressing estrogen production. Antiestrogens are one of three types of sex hormone antagonists, the others being antiandrogens and antiprogestogens. Antiestrogens are commonly used to stop steroid hormones, estrogen, from binding to the estrogen receptors leading to the decrease of estrogen levels. Decreased levels of estrogen can lead to complications in sexual development. Antiandrogens are sex hormone antagonists which are able to lower the production and the effects that testosterone can have on female bodies.

Afimoxifene, also known as 4-hydroxytamoxifen (4-OHT) and by its tentative brand name TamoGel, is a selective estrogen receptor modulator (SERM) of the triphenylethylene group and an active metabolite of tamoxifen. The drug is under development under the tentative brand name TamoGel as a topical gel for the treatment of hyperplasia of the breast. It has completed a phase II clinical trial for cyclical mastalgia, but further studies are required before afimoxifene can be approved for this indication and marketed.

Arzoxifene is a selective estrogen receptor modulator (SERM) of the benzothiophene group which was never marketed. It is a potent estrogen antagonist in mammary and uterine tissue while acting as an estrogen agonist to maintain bone density and lower serum cholesterol. Arzoxifene is a highly effective agent for prevention of mammary cancer induced in the rat by the carcinogen nitrosomethylurea and is significantly more potent than raloxifene in this regard. Arzoxifene is devoid of the uterotrophic effects of tamoxifen, suggesting that, in contrast to tamoxifen, it is unlikely that the clinical use of arzoxifene will increase the risk of developing endometrial carcinoma.
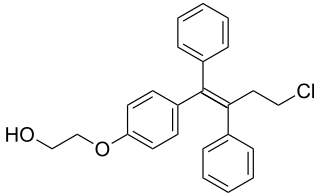
Ospemifene is an oral medication indicated for the treatment of dyspareunia – pain during sexual intercourse – encountered by some women, more often in those who are post-menopausal. Ospemifene is a selective estrogen receptor modulator (SERM) acting similarly to an estrogen on the vaginal epithelium, building vaginal wall thickness which in turn reduces the pain associated with dyspareunia. Dyspareunia is most commonly caused by "vulvar and vaginal atrophy."

Triphenylethylene (TPE) is a simple aromatic hydrocarbon that possesses weak estrogenic activity. Its estrogenic effects were discovered in 1937. TPE was derived from structural modification of the more potent estrogen diethylstilbestrol, which is a member of the stilbestrol group of nonsteroidal estrogens.

Ethamoxytriphetol is a synthetic nonsteroidal antiestrogen that was studied clinically in the late 1950s and early 1960s but was never marketed. MER-25 was first reported in 1958, and was the first antiestrogen to be discovered. It has been described as "essentially devoid of estrogenic activity" and as having "very low estrogenic activity in all species tested". However, some estrogenic effects in the uterus have been observed, so it is not a pure antiestrogen but is, instead, technically a selective estrogen receptor modulator (SERM). For all intents and purposes, it is a nearly pure antiestrogen, however.

Brilanestrant (INN) is a nonsteroidal combined selective estrogen receptor modulator (SERM) and selective estrogen receptor degrader (SERD) that was discovered by Aragon Pharmaceuticals and was under development by Genentech for the treatment of locally advanced or metastatic estrogen receptor (ER)-positive breast cancer.

Etacstil is an orally active, nonsteroidal, combined selective estrogen receptor modulator (SERM) and selective estrogen receptor degrader (SERD) that was developed for the treatment of estrogen receptor-positive breast cancer. It was shown to overcome antiestrogen resistance in breast cancer by altering the shape of the estrogen receptor, thus exhibiting SERD properties. Etacstil is a tamoxifen derivative and one of the first drugs to overcome tamoxifen-resistance. It is the predecessor of GW-7604, of which etacstil is a prodrug. This is analogous to the case of tamoxifen being a prodrug of afimoxifene (4-hydroxytamoxifen).

Droloxifene, also known as 3-hydroxytamoxifen, is a nonsteroidal selective estrogen receptor modulator (SERM) of the triphenylethylene group that was developed originally in Germany and later in Japan for the treatment of breast cancer, osteoporosis in men and postmenopausal women, and cardiovascular disorders but was abandoned and never marketed. It reached phase II and phase III clinical trials for these indications before development was discontinued in 2000. The drug was found to be significantly less effective than tamoxifen in the treatment of breast cancer in two phase III clinical trials.

Norendoxifen, also known as 4-hydroxy-N,N-didesmethyltamoxifen, is a nonsteroidal aromatase inhibitor (AI) of the triphenylethylene group that was never marketed. It is an active metabolite of the selective estrogen receptor modulator (SERM) tamoxifen. Unlike tamoxifen, norendoxifen is not a SERM, and instead has been found to act as a potent and selective competitive inhibitor of aromatase (Ki = 35 nM). Drugs with dual SERM and AI activity, such as 4'-hydroxynorendoxifen, have been developed from norendoxifen, and may have therapeutic potential as antiestrogens in the treatment of estrogen receptor-positive breast cancer.

Pipendoxifene (INN) is a nonsteroidal selective estrogen receptor modulator (SERM) that was under development by Ligand Pharmaceuticals and Wyeth-Ayerst Laboratories for the treatment of breast cancer but was not marketed. It is a member of the 2-phenylindole group of SERMs and is structurally related to zindoxifene and the marketed bazedoxifene. The drug reached phase II clinical trials before its development was discontinued. It was synthesized at the same time as bazedoxifene and was intended as a backup drug for bazedoxifene, only to be developed further if bazedoxifene had failed in clinical trials. No further development was reported after 2002 and it was formally announced that development had been terminated in November 2005.
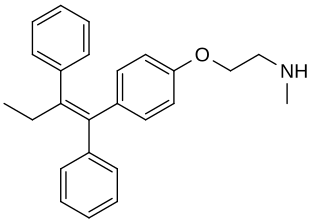
N-Desmethyltamoxifen is a major metabolite of tamoxifen, a selective estrogen receptor modulator (SERM). N-Desmethyltamoxifen is further metabolized into endoxifen (4-hydroxy-N-desmethyltamoxifen), which is thought to be the major active form of tamoxifen in the body. In one study, N-desmethyltamoxifen had an affinity for the estrogen receptor of 2.4% relative to estradiol. For comparison, tamoxifen, endoxifen, and afimoxifene (4-hydroxytamoxifen) had relative binding affinities of 2.8%, 181%, and 181%, respectively.