Calcium channel, voltage-dependent, T type, alpha 1H subunit, also known as CACNA1H, is a protein which in humans is encoded by the CACNA1H gene. [5] [6] [7]
Calcium channel, voltage-dependent, T type, alpha 1H subunit, also known as CACNA1H, is a protein which in humans is encoded by the CACNA1H gene. [5] [6] [7]
This gene encodes Cav3.2, a T-type member of the α1 subunit family, a protein in the voltage-dependent calcium channel complex. Calcium channels mediate the influx of calcium ions into the cell upon membrane polarization and consist of a complex of α1, α2δ, β, and γ subunits in a 1:1:1:1 ratio. The α1 subunit has 24 transmembrane segments and forms the pore through which ions pass into the cell. There are multiple isoforms of each of the proteins in the complex, either encoded by different genes or the result of alternative splicing of transcripts. Alternate transcriptional splice variants, encoding different isoforms, have been characterized for the gene described here. [5]
Studies suggest certain mutations in this gene lead to childhood absence epilepsy (CAE). [8] Variants of Cav3.2 with increased channel activity contribute to susceptibility to idiopathic generalized epilepsy (IGE), but are not sufficient to induce epilepsy on their own. [9] The SFARIgene database lists CACNA1H with an autism score of 2.1, indicating a candidate causal relationship with autism.
Voltage-gated calcium channels (VGCCs), also known as voltage-dependent calcium channels (VDCCs), are a group of voltage-gated ion channels found in the membrane of excitable cells (e.g., muscle, glial cells, neurons, etc.) with a permeability to the calcium ion Ca2+. These channels are slightly permeable to sodium ions, so they are also called Ca2+–Na+ channels, but their permeability to calcium is about 1000-fold greater than to sodium under normal physiological conditions.

Calcium channel, voltage-dependent, L type, alpha 1C subunit is a protein that in humans is encoded by the CACNA1C gene. Cav1.2 is a subunit of L-type voltage-dependent calcium channel.
T-type calcium channels are low voltage activated calcium channels that become inactivated during cell membrane hyperpolarization but then open to depolarization. The entry of calcium into various cells has many different physiological responses associated with it. Within cardiac muscle cell and smooth muscle cells voltage-gated calcium channel activation initiates contraction directly by allowing the cytosolic concentration to increase. Not only are T-type calcium channels known to be present within cardiac and smooth muscle, but they also are present in many neuronal cells within the central nervous system. Different experimental studies within the 1970s allowed for the distinction of T-type calcium channels from the already well-known L-type calcium channels. The new T-type channels were much different from the L-type calcium channels due to their ability to be activated by more negative membrane potentials, had small single channel conductance, and also were unresponsive to calcium antagonist drugs that were present. These distinct calcium channels are generally located within the brain, peripheral nervous system, heart, smooth muscle, bone, and endocrine system.
The R-type calcium channel is a type of voltage-dependent calcium channel. Like the others of this class, the α1 subunit forms the pore through which calcium enters the cell and determines most of the channel's properties. This α1 subunit is also known as the calcium channel, voltage-dependent, R type, alpha 1E subunit (CACNA1E) or Cav2.3 which in humans is encoded by the CACNA1E gene. They are strongly expressed in cortex, hippocampus, striatum, amygdala and interpeduncular nucleus.
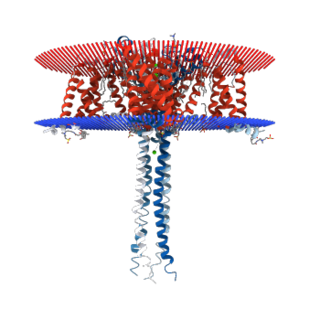
N-type calcium channels also called Cav2.2 channels are voltage gated calcium channels that are localized primarily on the nerve terminals and dendrites as well as neuroendocrine cells. The calcium N-channel consists of several subunits: the primary subunit α1B and the auxiliary subunits α2δ and β. The α1B subunit forms the pore through which the calcium enters and helps to determine most of the channel's properties. These channels play an important role in the neurotransmission during development. In the adult nervous system, N-type calcium channels are critically involved in the release of neurotransmitters, and in pain pathways. N-type calcium channels are the target of ziconotide, the drug prescribed to relieve intractable cancer pain. There are many known N-type calcium channel blockers that function to inhibit channel activity, although the most notable blockers are ω-conotoxins.

The L-type calcium channel is part of the high-voltage activated family of voltage-dependent calcium channel. "L" stands for long-lasting referring to the length of activation. This channel has four isoforms: Cav1.1, Cav1.2, Cav1.3, and Cav1.4.

Cav2.1, also called the P/Q voltage-dependent calcium channel, is a calcium channel found mainly in the brain. Specifically, it is found on the presynaptic terminals of neurons in the brain and cerebellum. Cav2.1 plays an important role in controlling the release of neurotransmitters between neurons. It is composed of multiple subunits, including alpha-1, beta, alpha-2/delta, and gamma subunits. The alpha-1 subunit is the pore-forming subunit, meaning that the calcium ions flow through it. Different kinds of calcium channels have different isoforms (versions) of the alpha-1 subunit. Cav2.1 has the alpha-1A subunit, which is encoded by the CACNA1A gene. Mutations in CACNA1A have been associated with various neurologic disorders, including familial hemiplegic migraine, episodic ataxia type 2, and spinocerebellar ataxia type 6.

Cav1.4 also known as the calcium channel, voltage-dependent, L type, alpha 1F subunit (CACNA1F), is a human gene.

Cav1.1 also known as the calcium channel, voltage-dependent, L type, alpha 1S subunit, (CACNA1S), is a protein which in humans is encoded by the CACNA1S gene. It is also known as CACNL1A3 and the dihydropyridine receptor.
Voltage-dependent L-type calcium channel subunit beta-2 is a protein that in humans is encoded by the CACNB2 gene.

Voltage-dependent L-type calcium channel subunit beta-4 is a protein that in humans is encoded by the CACNB4 gene.

Voltage-dependent L-type calcium channel subunit beta-1 is a protein that in humans is encoded by the CACNB1 gene.

Calcium channel, voltage-dependent, L type, alpha 1D subunit is a protein that in humans is encoded by the CACNA1D gene. Cav1.3 channels belong to the Cav1 family, which form L-type calcium currents and are sensitive to selective inhibition by dihydropyridines (DHP).

Voltage-dependent L-type calcium channel subunit beta-3 is a protein that in humans is encoded by the CACNB3 gene.
Calcium-activated potassium channel subunit beta-2 is a protein that in humans is encoded by the KCNMB2 gene.

Calcium-activated potassium channel subunit beta-3 is a protein that in humans is encoded by the KCNMB3 gene.

Voltage-dependent calcium channel gamma-3 subunit is a protein that in humans is encoded by the CACNG3 gene.

Calcium channel, voltage-dependent, T type, alpha 1I subunit, also known as CACNA1I or Cav3.3 is a protein which in humans is encoded by the CACNA1I gene.

The voltage-dependent N-type calcium channel subunit alpha-1B is a protein that in humans is encoded by the CACNA1B gene. The α1B protein, together with β and α2δ subunits forms N-type calcium channel PMID 26386135. It is a R-type calcium channel.

Calcium channel, voltage-dependent, T type, alpha 1G subunit, also known as CACNA1G or Cav3.1 is a protein which in humans is encoded by the CACNA1G gene. It is one of the primary targets in the pharmacology of absence seizure.
This article incorporates text from the United States National Library of Medicine, which is in the public domain.