
5-HT receptors, 5-hydroxytryptamine receptors, or serotonin receptors, are a group of G protein-coupled receptor and ligand-gated ion channels found in the central and peripheral nervous systems. They mediate both excitatory and inhibitory neurotransmission. The serotonin receptors are activated by the neurotransmitter serotonin, which acts as their natural ligand.

Noradrenergic and specific serotonergic antidepressants (NaSSAs) are a class of psychiatric drugs used primarily as antidepressants. They act by antagonizing the α2-adrenergic receptor and certain serotonin receptors such as 5-HT2A and 5-HT2C, but also 5-HT3, 5-HT6, and/or 5-HT7 in some cases. By blocking α2-adrenergic autoreceptors and heteroreceptors, NaSSAs enhance adrenergic and serotonergic neurotransmission in the brain involved in mood regulation, notably 5-HT1A-mediated transmission. In addition, due to their blockade of certain serotonin receptors, serotonergic neurotransmission is not facilitated in unwanted areas, which prevents the incidence of many side effects often associated with selective serotonin reuptake inhibitor (SSRI) antidepressants; hence, in part, the "specific serotonergic" label of NaSSAs.

Mesulergine (INNTooltip International Nonproprietary Name) (developmental code name CU-32085) is a drug of the ergoline group which was never marketed. It acts on serotonin and dopamine receptors. Specifically, it is an agonist of dopamine D2-like receptors and serotonin 5-HT6 receptors and an antagonist of serotonin 5-HT2A, 5-HT2B, 5-HT2C, and 5-HT7 receptors.. It also has affinity for the 5-HT1A, 5-HT1B, 5-HT1D, 5-HT1F, and 5-HT5A receptors. The compound had entered clinical trials for the treatment of Parkinson's disease; however, further development was halted due to adverse histological abnormalities in rats. It was also investigated for the treatment of hyperprolactinemia (high prolactin levels).

5-Hydroxytryptamine (serotonin) receptor 5A, also known as HTR5A, is a protein that in humans is encoded by the HTR5A gene. Agonists and antagonists for 5-HT receptors, as well as serotonin uptake inhibitors, present promnesic (memory-promoting) and/or anti-amnesic effects under different conditions, and 5-HT receptors are also associated with neural changes.

The 5HT6 receptor is a subtype of 5HT receptor that binds the endogenous neurotransmitter serotonin (5-hydroxytryptamine, 5HT). It is a G protein-coupled receptor (GPCR) that is coupled to Gs and mediates excitatory neurotransmission. HTR6 denotes the human gene encoding for the receptor.

The 5-HT7 receptor is a member of the GPCR superfamily of cell surface receptors and is activated by the neurotransmitter serotonin (5-hydroxytryptamine, 5-HT). The 5-HT7 receptor is coupled to Gs (stimulates the production of the intracellular signaling molecule cAMP) and is expressed in a variety of human tissues, particularly in the brain, the gastrointestinal tract, and in various blood vessels. This receptor has been a drug development target for the treatment of several clinical disorders. The 5-HT7 receptor is encoded by the HTR7 gene, which in humans is transcribed into 3 different splice variants.
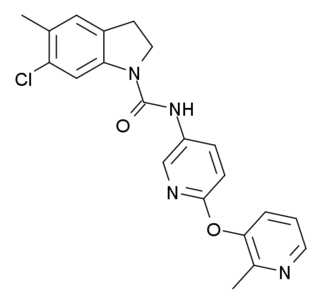
SB-242084 is a selective antagonist of the serotonin 5-HT2C receptor which is used in scientific research.

5-Carboxamidotryptamine (5-CT) is a tryptamine derivative closely related to the neurotransmitter serotonin.

2-Ethyl-5-methoxy-N,N-dimethyltryptamine (EMDT) is a tryptamine derivative which is used in scientific research. It acts as a selective 5-HT6 receptor agonist, with a Ki of 16 nM, and was one of the first selective agonists developed for this receptor. EMDT inhibits both short- and long-term memory formation in animal studies, and this effect can be reversed by the selective 5-HT6 antagonist SB-399,885. Additionally, it is active in the tail suspension test, suggesting that it could be an effective antidepressant.

SB-258585 is a drug which is used in scientific research. It acts as a potent, selective and orally active 5-HT6 receptor antagonist, with a Ki of 8.9nM. It is used in its 125I radiolabelled form to map the distribution of 5-HT6 receptors in the brain.

SB-399885 is a drug which is used in scientific research. It acts as a potent, selective and orally active 5-HT6 receptor antagonist, with a Ki of 9.0nM. SB-399885 and other 5-HT6 antagonists show nootropic effects in animal studies, as well as antidepressant and anxiolytic effects which are comparable to and synergistic with drugs such as imipramine and diazepam, and have been proposed as potential novel treatments for cognitive disorders such as schizophrenia and Alzheimer's disease.

SB-357134 is a drug which is used in scientific research. It acts as a potent, selective and orally active 5-HT6 receptor antagonist. SB-357134 and other 5-HT6 antagonists show nootropic effects in animal studies, and have been proposed as potential novel treatments for cognitive disorders such as schizophrenia and Alzheimer's disease.

SB-271046 is a drug which is used in scientific research. It was one of the first selective 5-HT6 receptor antagonists to be discovered, and was found through high-throughput screening of the SmithKline Beecham Compound Bank using cloned 5-HT6 receptors as a target, with an initial lead compound being developed into SB-271046 through a structure-activity relationship (SAR) study. SB-271046 was found to be potent and selective in vitro and had good oral bioavailability in vivo, but had poor penetration across the blood–brain barrier, so further SAR work was then conducted, which led to improved 5-HT6 antagonists such as SB-357,134 and SB-399,885.

SB-269970 is a drug and research chemical developed by GlaxoSmithKline used in scientific studies. It is believed to act as a selective 5-HT7 receptor antagonist (EC50 = 1.25 nM) (or possibly inverse agonist). A subsequent study in guinea pig at a concentration of 10 μM showed that it also blocks the α2-adrenergic receptor. The large difference in test concentrations however confirms the selectivity of SB-269970 for the 5-HT7 receptor.
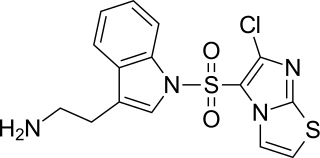
WAY-181187 is a high affinity and selective 5-HT6 receptor full agonist. It induces robust increases in extracellular GABA levels in the frontal cortex, hippocampus, striatum, and amygdala of rats without affecting concentrations in the nucleus accumbens or thalamus, and has modest to no effects on norepinephrine, serotonin, dopamine, or glutamate levels in these areas. WAY-181187 has demonstrated preclinical efficacy in rodent models of depression, anxiety, and notably obsessive-compulsive disorder, though it has also been shown to impair cognition and memory.
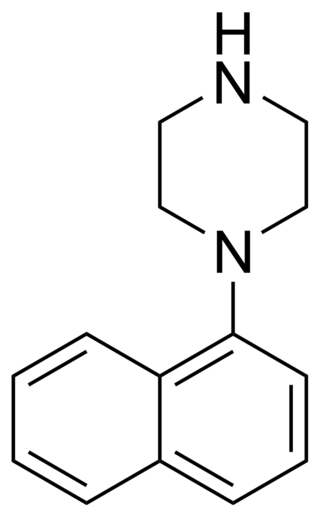
1-(1-Naphthyl)piperazine (1-NP) is a drug which is a phenylpiperazine derivative. It acts as a non-selective, mixed serotonergic agent, exerting partial agonism at the 5-HT1A, 5-HT1B, 5-HT1D, 5-HT1E, and 5-HT1F receptors, while antagonizing the 5-HT2A, 5-HT2B, and 5-HT2C receptors. It has also been shown to possess high affinity for the 5-HT3, 5-HT5A, 5-HT6, and 5-HT7 receptors, and may bind to 5-HT4 and the SERT as well. In animals it produces effects including hyperphagia, hyperactivity, and anxiolysis, of which are all likely mediated predominantly or fully by blockade of the 5-HT2C receptor.
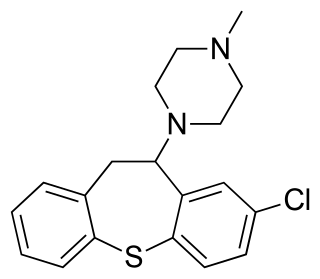
Clorotepine, also known as octoclothepin or octoclothepine, is an antipsychotic of the tricyclic group which was derived from perathiepin in 1965 and marketed in the Czech Republic by Spofa in or around 1971 for the treatment of schizophrenic psychosis.

ST-1936 (2-methyl-5-chloro-N,N-dimethyltryptamine) is a tryptamine derivative which is used in scientific research. It acts as a selective 5-HT6 receptor agonist, with a Ki of 13 nM, and much weaker action at 5-HT2B and 5-HT7 subtypes. In animal studies it has been found to increase dopamine and noradrenaline mediated signalling but decreases glutamatergic transmission, and has antidepressant effects.

Amesergide is a serotonin receptor antagonist of the ergoline and lysergamide families related to methysergide which was under development by Eli Lilly and Company for the treatment of a variety of conditions including depression, anxiety, schizophrenia, male sexual dysfunction, migraine, and thrombosis but was never marketed. It reached phase II clinical trials for the treatment of depression, erectile dysfunction, and premature ejaculation prior to the discontinuation of its development.

JNJ-18038683 is a potent and selective antagonist of the 5HT7 serotonin receptor discovered by Johnson & Johnson. It has nootropic and antidepressant effects in both animal and human studies and has progressed to Phase II trials as an adjunctive treatment for improving cognition and mood in stable bipolar disorder; it has been found to reduce REM sleep (the lightest stage of sleep, elevated in depression) in humans and block circadian rhythm phase-shift advances in mice.




















