
5-HT receptors, 5-hydroxytryptamine receptors, or serotonin receptors, are a group of G protein-coupled receptor and ligand-gated ion channels found in the central and peripheral nervous systems. They mediate both excitatory and inhibitory neurotransmission. The serotonin receptors are activated by the neurotransmitter serotonin, which acts as their natural ligand.

SB-277,011A is a drug which acts as a potent and selective dopamine D3 receptor antagonist, which is around 80–100 times selective for D3 over D2, and lacks any partial agonist activity.

Orexin receptor type 2 (Ox2R or OX2), also known as hypocretin receptor type 2 (HcrtR2), is a protein that in humans is encoded by the HCRTR2 gene. It should not be confused for the protein CD200R1 which shares the alias OX2R but is a distinct, unrelated gene located on the human chromosome 3.
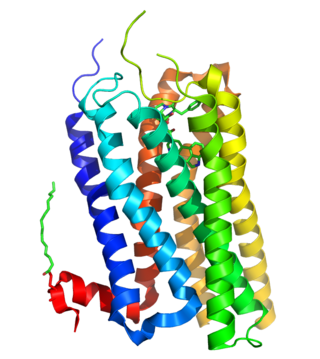
5-Hydroxytryptamine receptor 2B (5-HT2B) also known as serotonin receptor 2B is a protein that in humans is encoded by the HTR2B gene. 5-HT2B is a member of the 5-HT2 receptor family that binds the neurotransmitter serotonin (5-hydroxytryptamine, 5-HT). Like all 5-HT2 receptors, the 5-HT2B receptor is Gq/G11-protein coupled, leading to downstream activation of phospholipase C.

5-Hydroxytryptamine (serotonin) receptor 5A, also known as HTR5A, is a protein that in humans is encoded by the HTR5A gene. Agonists and antagonists for 5-HT receptors, as well as serotonin uptake inhibitors, present promnesic (memory-promoting) and/or anti-amnesic effects under different conditions, and 5-HT receptors are also associated with neural changes.

The 5HT6 receptor is a subtype of 5HT receptor that binds the endogenous neurotransmitter serotonin (5-hydroxytryptamine, 5HT). It is a G protein-coupled receptor (GPCR) that is coupled to Gs and mediates excitatory neurotransmission. HTR6 denotes the human gene encoding for the receptor.

Dopamine receptor D3 is a protein that in humans is encoded by the DRD3 gene.

The 5-HT7 receptor is a member of the GPCR superfamily of cell surface receptors and is activated by the neurotransmitter serotonin (5-hydroxytryptamine, 5-HT). The 5-HT7 receptor is coupled to Gs (stimulates the production of the intracellular signaling molecule cAMP) and is expressed in a variety of human tissues, particularly in the brain, the gastrointestinal tract, and in various blood vessels. This receptor has been a drug development target for the treatment of several clinical disorders. The 5-HT7 receptor is encoded by the HTR7 gene, which in humans is transcribed into 3 different splice variants.

CGP-37849 is a competitive antagonist at the NMDA receptor. It is a potent, orally active anticonvulsant in animal models, and was researched for the treatment of epilepsy. It also has neuroprotective activity and shows antidepressant and anxiolytic effects.

SB-258585 is a drug which is used in scientific research. It acts as a potent, selective and orally active 5-HT6 receptor antagonist, with a Ki of 8.9nM. It is used in its 125I radiolabelled form to map the distribution of 5-HT6 receptors in the brain.

SB-399885 is a drug which is used in scientific research. It acts as a potent, selective and orally active 5-HT6 receptor antagonist, with a Ki of 9.0nM. SB-399885 and other 5-HT6 antagonists show nootropic effects in animal studies, as well as antidepressant and anxiolytic effects which are comparable to and synergistic with drugs such as imipramine and diazepam, and have been proposed as potential novel treatments for cognitive disorders such as schizophrenia and Alzheimer's disease.

SB-271046 is a drug which is used in scientific research. It was one of the first selective 5-HT6 receptor antagonists to be discovered, and was found through high-throughput screening of the SmithKline Beecham Compound Bank using cloned 5-HT6 receptors as a target, with an initial lead compound being developed into SB-271046 through a structure-activity relationship (SAR) study. SB-271046 was found to be potent and selective in vitro and had good oral bioavailability in vivo, but had poor penetration across the blood–brain barrier, so further SAR work was then conducted, which led to improved 5-HT6 antagonists such as SB-357,134 and SB-399,885.
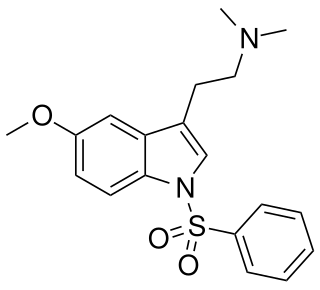
MS-245 is a tryptamine derivative used in scientific research. It acts as a selective 5-HT6 receptor antagonist with a Ki of 2.3 nM, and was derived through structure-activity relationship development of the selective 5-HT6 agonist EMDT. It has been used as a lead compound for further development of tryptamine-derived 5-HT6 antagonists. In animal studies it has been shown to boost the activity of, but not substitute for, both amphetamine and nicotine.

RS-102221 is a drug developed by Hoffmann–La Roche, which was one of the first compounds discovered that acts as a potent and selective antagonist at the serotonin 5-HT2C receptor, with around 100× selectivity over the closely related 5-HT2A and 5-HT2B receptors. It has anxiolytic effects in animal studies, increases the effectiveness of SSRI antidepressants, and shows a complex interaction with cocaine, increasing some effects but decreasing others, reflecting a role for the 5-HT2C receptor in regulation of the dopamine signalling system in the brain.

SB-269970 is a drug and research chemical developed by GlaxoSmithKline used in scientific studies. It is believed to act as a selective 5-HT7 receptor antagonist (EC50 = 1.25 nM) (or possibly inverse agonist). A subsequent study in guinea pig at a concentration of 10 μM showed that it also blocks the α2-adrenergic receptor. The large difference in test concentrations however confirms the selectivity of SB-269970 for the 5-HT7 receptor.

LY-379,268 is a drug that is used in neuroscience research, which acts as a potent and selective agonist for the group II metabotropic glutamate receptors (mGluR2/3).
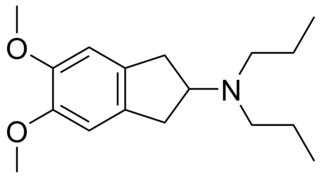
PNU-99,194(A) (or U-99,194(A)) is a drug which acts as a moderately selective D3 receptor antagonist with ~15-30-fold preference for D3 over the D2 subtype. Though it has substantially greater preference for D3 over D2, the latter receptor does still play some role in its effects, as evidenced by the fact that PNU-99,194 weakly stimulates both prolactin secretion and striatal dopamine synthesis, actions it does not share with the more selective (100-fold) D3 receptor antagonists S-14,297 and GR-103,691.

SB-206553 is a drug which acts as a mixed antagonist for the 5-HT2B and 5-HT2C serotonin receptors. It has anxiolytic properties in animal studies and interacts with a range of other drugs. It has also been shown to act as a positive allosteric modulator of α7 nicotinic acetylcholine receptors. Modified derivatives of SB-206553 have been used to probe the structure of the 5-HT2B receptor.

Aticaprant, also known by its developmental codes JNJ-67953964, CERC-501, and LY-2456302, is a κ-opioid receptor (KOR) antagonist which is under development for the treatment of major depressive disorder. A regulatory application for approval of the medication is expected to be submitted by 2025. Aticaprant is taken by mouth.

SB-705498 is a drug which acts as a potent and selective blocker of the TRPV1 ion channel. It has been evaluated in clinical trials for the treatment of rhinitis and chronic cough.




















