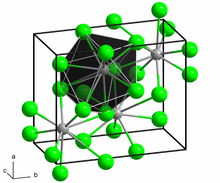
Titanium(III) chloride is the inorganic compound with the formula TiCl3. At least four distinct species have this formula; additionally hydrated derivatives are known. TiCl3 is one of the most common halides of titanium and is an important catalyst for the manufacture of polyolefins.

Titanium(II) chloride is the chemical compound with the formula TiCl2. The black solid has been studied only moderately, probably because of its high reactivity. Ti(II) is a strong reducing agent: it has a high affinity for oxygen and reacts irreversibly with water to produce H2. The usual preparation is the thermal disproportionation of TiCl3 at 500 °C. The reaction is driven by the loss of volatile TiCl4:

Samarium(II) chloride (SmCl2) is a chemical compound, used as a radical generating agent in the ketone-mediated intraannulation reaction.
Magnesium compounds are compounds formed by the element magnesium (Mg). These compounds are important to industry and biology, including magnesium carbonate, magnesium chloride, magnesium citrate, magnesium hydroxide, magnesium oxide, magnesium sulfate, and magnesium sulfate heptahydrate.

Indium(III) bromide, (indium tribromide), InBr3, is a chemical compound of indium and bromine. It is a Lewis acid and has been used in organic synthesis.
There are three sets of Indium halides, the trihalides, the monohalides, and several intermediate halides. In the monohalides the oxidation state of indium is +1 and their proper names are indium(I) fluoride, indium(I) chloride, indium(I) bromide and indium(I) iodide.

Zirconium(III) chloride is an inorganic compound with formula ZrCl3. It is a blue-black solid that is highly sensitive to air.
The nitridoborates are chemical compounds of boron and nitrogen with metals. These compounds are typically produced at high temperature by reacting hexagonal boron nitride with metal nitrides or by metathesis reactions involving nitridoborates. A wide range of these compounds have been made involving lithium, alkaline earth metals and lanthanides, and their structures determined using crystallographic techniques such as X-ray crystallography. Structurally one of their interesting features is the presence of polyatomic anions of boron and nitrogen where the geometry and the B–N bond length have been interpreted in terms of π-bonding.
Cobalt(III) chloride or cobaltic chloride is an unstable and elusive compound of cobalt and chlorine with formula CoCl
3. In this compound, the cobalt atoms have a formal charge of +3.
The inorganic imides are compounds containing an ion composed of nitrogen bonded to hydrogen with formula HN2−. Organic imides have the NH group, and two single or one double covalent bond to other atoms. The imides are related to the inorganic amides (H2N−), the nitrides (N3−) and the nitridohydrides (N3−•H−).
A chloride nitride is a mixed anion compound containing both chloride (Cl−) and nitride ions (N3−). Another name is metallochloronitrides. They are a subclass of halide nitrides or pnictide halides.
Arsenide bromides or bromide arsenides are compounds containing anions composed of bromide (Br−) and arsenide (As3−). They can be considered as mixed anion compounds. They are in the category of pnictidehalides. Related compounds include the arsenide chlorides, arsenide iodides, phosphide bromides, and antimonide bromides.
An iodide nitride is a mixed anion compound containing both iodide (I−) and nitride ions (N3−). Another name is metalloiodonitrides. They are a subclass of halide nitrides or pnictide halides. Some different kinds include ionic alkali or alkaline earth salts, small clusters where metal atoms surround a nitrogen atom, layered group 4 element 2-dimensional structures, and transition metal nitrido complexes counter-balanced with iodide ions. There is also a family with rare earth elements and nitrogen and sulfur in a cluster.
Carbide chlorides are mixed anion compounds containing chloride anions and anions consisting entirely of carbon. In these compounds there is no bond between chlorine and carbon. But there is a bond between a metal and carbon. Many of these compounds are cluster compounds, in which metal atoms encase a carbon core, with chlorine atoms surrounding the cluster. The chlorine may be shared between clusters to form polymers or layers. Most carbon chloride compounds contain rare earth elements. Some are known from group 4 elements. The hexatungsten carbon cluster can be oxidised and reduced, and so have different numbers of chlorine atoms included.
Carbide bromides are mixed anion compounds containing bromide and carbide anions. Many carbide bromides are cluster compounds, containing on, two or more carbon atoms in a core, surrounded by a layer of metal atoms, encased in a shell of bromide ions. These ions may be shared between clusters to form chains, double chains or layers.
Carbide iodides are mixed anion compounds containing iodide and carbide anions. Many carbide iodides are cluster compounds, containing one, two or more carbon atoms in a core, surrounded by a layer of metal atoms, and encased in a shell of iodide ions. These ions may be shared between clusters to form chains, double chains or layers.
Germanide halides are compound that include the germanide (Ge4−) anion and a halide such as chloride (Cl−), bromide (Br−) or iodide (I−). They include germanide iodides, germanide bromides or germanide chlorides. They can be considered as mixed anion compounds. They are in the category of tetrelidehalides. Related compounds include the silicide iodides, and carbide iodides.
Antimonide iodides or iodide antimonides are compounds containing anions composed of iodide (I−) and antimonide (Sb3−). They can be considered as mixed anion compounds. They are in the category of pnictide halides. Related compounds include the antimonide chlorides, antimonide bromides, phosphide iodides, and arsenide iodides.

Europium compounds are compounds formed by the lanthanide metal europium (Eu). In these compounds, europium generally exhibits the +3 oxidation state, such as EuCl3, Eu(NO3)3 and Eu(CH3COO)3. Compounds with europium in the +2 oxidation state are also known. The +2 ion of europium is the most stable divalent ion of lanthanide metals in aqueous solution. Lipophilic europium complexes often feature acetylacetonate-like ligands, e.g., Eufod.
Lanthanide chlorides are a group of chemical compounds that can form between a lanthanide element and chlorine. The lanthanides in these compounds are usually in the +2 and +3 oxidation states, although compounds with lanthanides in lower oxidation states exist.