A progestogen, also referred to as a progestagen, gestagen, or gestogen, is a type of medication which produces effects similar to those of the natural female sex hormone progesterone in the body. A progestin is a synthetic progestogen. Progestogens are used most commonly in hormonal birth control and menopausal hormone therapy. They can also be used in the treatment of gynecological conditions, to support fertility and pregnancy, to lower sex hormone levels for various purposes, and for other indications. Progestogens are used alone or in combination with estrogens. They are available in a wide variety of formulations and for use by many different routes of administration. Examples of progestogens include natural or bioidentical progesterone as well as progestins such as medroxyprogesterone acetate and norethisterone.

Hormonal contraception refers to birth control methods that act on the endocrine system. Almost all methods are composed of steroid hormones, although in India one selective estrogen receptor modulator is marketed as a contraceptive. The original hormonal method—the combined oral contraceptive pill—was first marketed as a contraceptive in 1960. In the ensuing decades, many other delivery methods have been developed, although the oral and injectable methods are by far the most popular. Hormonal contraception is highly effective: when taken on the prescribed schedule, users of steroid hormone methods experience pregnancy rates of less than 1% per year. Perfect-use pregnancy rates for most hormonal contraceptives are usually around the 0.3% rate or less. Currently available methods can only be used by women; the development of a male hormonal contraceptive is an active research area.

The Concept Foundation is a non-profit foundation which was established by the UNDP/UNFPA/WHO/WB Special program in Reproductive Health (WHO/HRP), PATH, the World Bank in 1989 in Bangkok, Thailand, "as a mechanism through which WHO’s rights associated with an injectable contraceptive, Cyclofem, could be licensed to potential producers in developing countries". Estradiol cypionate/medroxyprogesterone acetate, is a once-a-month combined injectable contraceptive which contains 25 mg of medroxyprogesterone acetate—the same ingredient in Depo Provera—and 5 mg of estradiol cypionate.
Combined injectable contraceptives (CICs) are a form of hormonal birth control for women. They consist of monthly injections of combined formulations containing an estrogen and a progestin to prevent pregnancy.

Algestone acetophenide, also known more commonly as dihydroxyprogesterone acetophenide (DHPA) and sold under the brand names Perlutal and Topasel among others, is a progestin medication which is used in combination with an estrogen as a form of long-lasting injectable birth control. It has also been used alone, but is no longer available as a standalone medication. DHPA is not active by mouth and is given once a month by injection into muscle.

Medroxyprogesterone acetate (MPA), also known as depot medroxyprogesterone acetate (DMPA) in injectable form and sold under the brand name Depo-Provera among others, is a hormonal medication of the progestin type. It is used as a method of birth control and as a part of menopausal hormone therapy. It is also used to treat endometriosis, abnormal uterine bleeding, paraphilia, and certain types of cancer. The medication is available both alone and in combination with an estrogen. It is taken by mouth, used under the tongue, or by injection into a muscle or fat.

Estradiol cypionate (EC), sold under the brand name Depo-Estradiol among others, is an estrogen medication which is used in hormone therapy for menopausal symptoms and low estrogen levels in women, in hormone therapy for trans women, and in hormonal birth control for women. It is given by injection into muscle once every 1 to 4 weeks.

Norethisterone enanthate (NETE), also known as norethindrone enanthate, is a form of hormonal birth control which is used to prevent pregnancy in women. It is used both as a form of progestogen-only injectable birth control and in combined injectable birth control formulations. It may be used following childbirth, miscarriage, or abortion. The failure rate per year in preventing pregnancy for the progestogen-only formulation is 2 per 100 women. Each dose of this form lasts two months with only up to two doses typically recommended.
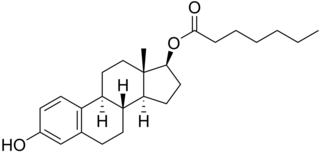
Estradiol enantate, also spelled estradiol enanthate and sold under the brand names Perlutal and Topasel among others, is an estrogen medication which is used in hormonal birth control for women. It is formulated in combination with dihydroxyprogesterone acetophenide, a progestin, and is used specifically as a combined injectable contraceptive. Estradiol enantate is not available for medical use alone. The medication, in combination with DHPA, is given by injection into muscle once a month.

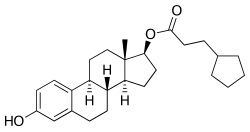
Estradiol hexahydrobenzoate (EHHB), sold under a number of brand names including Benzo-Ginoestril A.P., BenzoGynoestryl Retard, Ginestryl-15-Depot, Menodin, and Tardoginestryl, is an estrogen medication which was previously used for indications such as menopausal hormone therapy and gynecological disorders. EHHB is given by injection into muscle at regular intervals, for instance once every few weeks.
Progestogen-only injectable contraceptives (POICs) are a form of hormonal contraception and progestogen-only contraception that are administered by injection and providing long-lasting birth control. As opposed to combined injectable contraceptives, they contain only a progestogen without an estrogen, and include two progestin preparations:

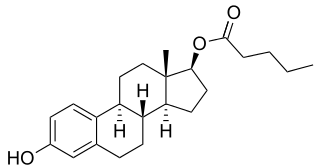
Estradiol benzoate butyrate (EBB), sold under the brand names Neolutin N, Redimen, Soluna, and Unijab and formerly known under the developmental code name Unimens, is an estrogen medication which is used in hormonal birth control for women. It is formulated in combination with dihydroxyprogesterone acetophenide, a progestin, and is used specifically as a combined injectable contraceptive. EBB is not available for medical use alone. The medication, in combination with DHPA, is given by injection into muscle once a month.
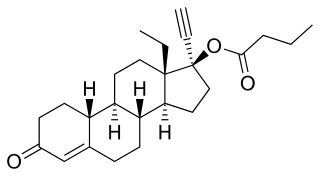
Levonorgestrel butanoate (LNG-B), or levonorgestrel 17β-butanoate, is a steroidal progestin of the 19-nortestosterone group which was developed by the World Health Organization (WHO) in collaboration with the Contraceptive Development Branch (CDB) of the National Institute of Child Health and Human Development as a long-acting injectable contraceptive. It is the C17β butanoate ester of levonorgestrel, and acts as a prodrug of levonorgestrel in the body. The drug is at or beyond the phase III stage of clinical development, but has not been marketed at this time. It was first described in the literature, by the WHO, in 1983, and has been under investigation for potential clinical use since then.
Conjugated estrogens/medroxyprogesterone acetate (CEs/MPA), sold under the brand names Prempro and Premphase, is a combination product of conjugated equine estrogens (Premarin), an estrogen collected from horse urine, and medroxyprogesterone acetate (Provera), a progestogen, which is used in menopausal hormone therapy for the treatment of menopausal symptoms.
Estradiol valerate/norethisterone enantate (EV/NETE), sold under the brand name Mesigyna among others, is a form of combined injectable birth control which is used to prevent pregnancy in women. It contains estradiol valerate (EV), an estrogen, and norethisterone enantate (NETE), a progestin. The medication is given once a month by injection into muscle.

Estradiol valerate/hydroxyprogesterone caproate (EV/OHPC), sold under the brand names Gravibinon and Injectable No. 1 among others, is a combined estrogen and progestogen medication which is used in the treatment of threatened miscarriage and other indications and as a form of combined injectable birth control to prevent pregnancy. It contains estradiol valerate (EV), an estrogen, and hydroxyprogesterone caproate (OHPC), a progestin. The medication is given by injection into muscle once a day to once a month depending on the indication.

Estradiol enantate/algestone acetophenide, also known as estradiol enantate/dihydroxyprogesterone acetophenide (E2-EN/DHPA) and sold under the brand names Perlutal and Topasel among others, is a form of combined injectable birth control which is used to prevent pregnancy. It contains estradiol enantate (E2-EN), an estrogen, and algestone acetophenide, a progestin. The medication is given once a month by injection into muscle.

Estradiol benzoate butyrate/algestone acetophenide, also known as estradiol benzoate butyrate/dihydroxyprogesterone acetophenide (EBB/DHPA) and sold under the brand names Neolutin N, Redimen, Soluna, and Unijab, is a form of combined injectable birth control which is used in Peru and Singapore. It contains estradiol benzoate butyrate (EBB), an estrogen, and algestone acetophenide, a progestin. The medication is given once per month by injection into muscle.
Menstrual suppression refers to the practice of using hormonal management to stop or reduce menstrual bleeding. In contrast to surgical options for this purpose, such as hysterectomy or endometrial ablation, hormonal methods to manipulate menstruation are reversible.