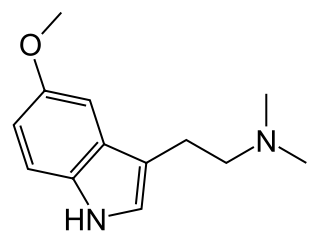
5-MeO-DMT (5-methoxy-N,N-dimethyltryptamine) or O-methyl-bufotenin is a psychedelic of the tryptamine class. It is found in a wide variety of plant species, and also is secreted by the glands of at least one toad species, the Colorado River toad. Like its close relatives DMT and bufotenin (5-HO-DMT), it has been used as an entheogen in South America. Slang terms include Five-methoxy, The power, and Toad venom.
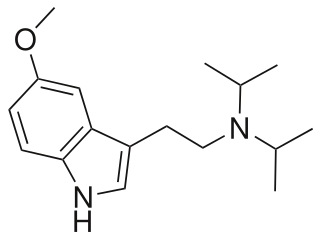
5-Methoxy-N,N-diisopropyltryptamine is a psychedelic tryptamine and the methoxy derivative of diisopropyltryptamine (DiPT).

5-MeO-MiPT is a psychedelic and hallucinogenic drug, used by some as an entheogen. It has structural and pharmacodynamic properties similar to the drugs 5-MeO-DiPT, DiPT, and MiPT. It is commonly used as a "substitute" for 5-MeO-DiPT because of the very similar structure and effects.

5-Fluoro-α-methyltryptamine, also known as PAL-544, is a putative stimulant, entactogen, and psychedelic tryptamine derivative related to α-methyltryptamine (αMT). It has been found to act as a well-balanced serotonin-norepinephrine-dopamine releasing agent, a 5-HT2A receptor agonist, and a potent and specific MAO-A inhibitor. which suggests that 5-fluoro-αMT could be an active psychedelic in humans, although it is not known to have been tested in humans and could be dangerous due to its strong inhibition of MAO-A.
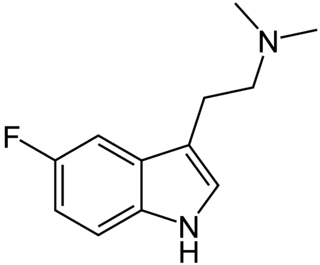
5-Fluoro-N,N-dimethyltryptamine is a tryptamine derivative related to compounds such as 5-bromo-DMT and 5-MeO-DMT. Fluorination of psychedelic tryptamines either reduces or has little effect on 5-HT2A/C receptor affinity or intrinsic activity, although 6-fluoro-DET is inactive as a psychedelic despite acting as a 5-HT2A agonist, while 4-fluoro-5-methoxy-DMT is a much stronger agonist at 5-HT1A than 5-HT2A.

Dimemebfe (5-MeO-BFE) is a recreational drug and research chemical. It acts as an agonist for the 5-HT1A and 5-HT2 family of serotonin receptors. It is related in structure to the psychedelic tryptamine derivative 5-MeO-DMT, but with the indole nitrogen replaced by oxygen, making dimemebfe a benzofuran derivative. It is several times less potent as a serotonin agonist than 5-MeO-DMT and with relatively more activity at 5-HT1A, but still shows strongest effects at the 5-HT2 family of receptors.

5-Methoxy-7,N,N-trimethyltryptamine (5-MeO-7,N,N-TMT, 5-MeO-7-TMT), is a tryptamine derivative which acts as an agonist at the 5-HT2 serotonin receptors. In animal tests, both 7,N,N-TMT and 5-MeO-7,N,N-TMT produced behavioural responses similar to those of psychedelic drugs such as DMT and 5-MeO-DMT, but compounds with larger 7-position substituents such as 7-ethyl-DMT and 7-bromo-DMT did not produce psychedelic-appropriate responding despite high 5-HT2 receptor binding affinity, suggesting these may be antagonists or weak partial agonists for the 5-HT2 receptors. The related compound 7-MeO-MiPT (cf. 5-MeO-MiPT) was also found to be inactive, suggesting that the 7-position has poor tolerance for bulky groups at this position, at least if agonist activity is desired.

7,N,N-trimethyltryptamine (7-methyl-DMT, 7-TMT), is a tryptamine derivative which acts as an agonist of 5-HT2 receptors. In animal tests, both 7-TMT and its 5-methoxy derivative 5-MeO-7-TMT produced behavioural responses similar to those of psychedelic drugs such as DMT, but the larger 7-ethyl and 7-bromo derivatives of DMT did not produce psychedelic responses despite having higher 5-HT2 receptor affinity in vitro (cf. DOBU, DOAM). 7-TMT also weakly inhibits reuptake of serotonin but with little effect on dopamine or noradrenaline reuptake.
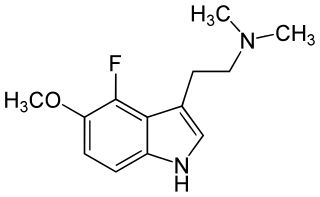
4-Fluoro-5-Methoxy-N,N-dimethyltryptamine (4-F-5-MeO-DMT) was first described by David E. Nichols team in 2000. It is a potent 5-HT1A agonist. Substitution with the 4-fluorine markedly increased 5-HT1A selectivity over 5-HT2A/2C receptors with potency greater than that of the 5-HT1A agonist 8-OH-DPAT.

O-4310 (1-isopropyl-6-fluoro-psilocin) is a tryptamine derivative developed by Organix Inc which acts as a serotonin receptor agonist. It is claimed to have an EC50 of 5nM at 5-HT2A with 89% efficacy vs 5-HT, and 100x selectivity over 5-HT2C, while being apparently inactive at the 5-HT2B antitarget.
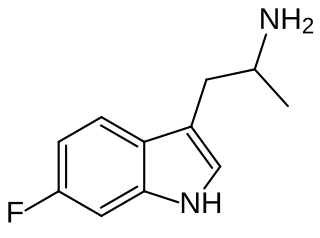
6-Fluoro-α-methyltryptamine is a tryptamine derivative related to compounds such as alpha-methyltryptamine and 5-MeO-AMT, which has been sold as a designer drug. Animal tests showed it to be somewhat less active than AMT or 5-fluoro-AMT, but it was nevertheless allegedly manufactured and sold from the laboratory operated by Leonard Pickard and Gordon Todd Skinner, who described 6-fluoro-AMT as "a beast".

5-Chloro-α-methyltryptamine (5-Chloro-αMT), also known as PAL-542, is a tryptamine derivative related to α-methyltryptamine (αMT) and one of only a few known specific serotonin-dopamine releasing agents (SDRAs). It has been investigated in animals as a potential treatment for cocaine dependence. The EC50 values of 5-chloro-αMT in evoking the in vitro release of serotonin (5-HT), dopamine (DA), and norepinephrine (NE) in rat synaptosomes were reported as 16 nM, 54 nM, and 3434 nM, with an NE/DA ratio of 63.6 and a DA/5-HT ratio of 3.38, indicating that it is a highly specific and well-balanced SDRA. However, 5-chloro-αMT has also been found to act as a potent full agonist of the 5-HT2A receptor, with an EC50 value of 6.27 nM and an efficacy of 105%, and almost assuredly acts as a potent agonist of other serotonin receptors as well.

5-MeO-DiBF is a psychedelic that has been sold online as a designer drug and was first definitively identified in December 2015 by a forensic laboratory in Slovenia. It is thought to act as an agonist for the 5-HT1A and 5-HT2 family of serotonin receptors. It is related in structure to the psychedelic tryptamine derivative 5-MeO-DiPT, but with the indole nitrogen replaced by oxygen, making 5-MeO-DiBF a benzofuran derivative. It is several times less potent as a serotonin agonist than 5-MeO-DiPT and with relatively more activity at 5-HT1A, but still shows strongest effects at the 5-HT2 family of receptors. LEGAL STATUS. It is not controlled under the 1971 Convention on Psychotropic Substances, so thus it has a legal grey area in many countries of the world, but its consumption still could be persecuted under severe analogue acts or the intend of sell to human consumption.

5-MeO-MET (5-Methoxy-N-methyl-N-ethyltryptamine) is a relatively rare designer drug from the substituted tryptamine family, related to compounds such as N-methyl-N-ethyltryptamine and 5-MeO-DMT. It was first synthesised in the 1960s and was studied to a limited extent, but was first identified on the illicit market in June 2012 in Sweden. It was made illegal in Norway in 2013, and is controlled under analogue provisions in numerous other jurisdictions.

7-Chloro-α-methyltryptamine (7-Cl-AMT) is a tryptamine derivative with stimulant effects, invented in the 1960s. It is a weak monoamine oxidase inhibitor but its pharmacology has not otherwise been studied by modern techniques, though several closely related compounds are known to act as serotonin–dopamine releasing agents and agonists of the 5-HT2A receptor.

5-Fluoro-α-ethyltryptamine (5-F-AET) is a tryptamine derivative which acts as a serotonin–dopamine releasing agent and agonist of the 5-HT2A receptor.
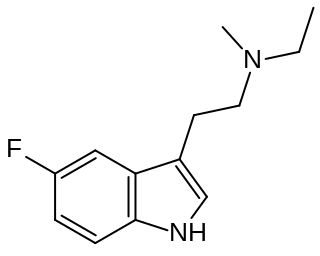
5-Fluoro-MET (5F-MET, 5-fluoro-N-methyl-N-ethyltryptamine) is a psychedelic tryptamine derivative related to drugs such as 5-Fluoro-DMT and N-Methyl-N-ethyltryptamine (MET). It acts as an agonist at the 5-HT2A receptor with an EC50 of 20.6 nM and produces a head-twitch response in animal studies. Ring fluorination in this case increases efficacy at 5-HT2A, with 5F-MET having an efficacy of 87.6% vs 5-HT, vs 36.2% for the partial agonist MET. It is claimed to have antidepressant activity.
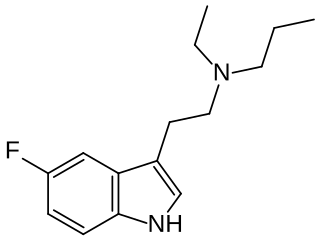
5-Fluoro-EPT (5F-EPT, 5-fluoro-N-ethyl-N-propyltryptamine) is a psychedelic tryptamine derivative related to drugs such as EPT and 5-MeO-EPT. It acts as a potent full agonist at the 5-HT2A receptor with an EC50 of 5.54 nM and an efficacy of 104% (compared to serotonin). It produces a head-twitch response in animal studies, and is claimed to have antidepressant activity.

5-Fluoro-DET is a tryptamine derivative related to drugs such as DET and 5-MeO-DET. It acts as an inhibitor of the enzyme myeloperoxidase, and is also thought to be an agonist at the 5-HT2A receptor.

5F-MPMI is a tryptamine derivative which acts as a serotonin receptor agonist, selective for the 5-HT2 subtypes but with similar affinity to all three receptors, having strongest activity at 5-HT2B and weakest at 5-HT2A.




















