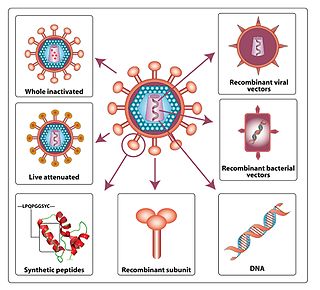
An HIV vaccine is a potential vaccine that could be either a preventive vaccine or a therapeutic vaccine, which means it would either protect individuals from being infected with HIV or treat HIV-infected individuals.

In immunology, seroconversion is the development of specific antibodies in the blood serum as a result of infection or immunization, including vaccination. During infection or immunization, antigens enter the blood, and the immune system begins to produce antibodies in response. Before seroconversion, the antigen itself may or may not be detectable, but the antibody is absent. During seroconversion, the antibody is present but not yet detectable. After seroconversion, the antibody is detectable by standard techniques and remains detectable unless the individual seroreverts. Seroreversion, or loss of antibody detectability, can occur due to weakening of the immune system or waning antibody concentration over time. Seroconversion refers the production of specific antibodies against specific antigens, meaning that a single infection could cause multiple waves of seroconversion against different antigens. Similarly, a single antigen could cause multiple waves of seroconversion with different classes of antibodies. For example, most antigens prompt seroconversion for the IgM class of antibodies first, and subsequently the IgG class.

CSL Limited is an Australian multinational specialty biotechnology company that researches, develops, manufactures, and markets products to treat and prevent serious human medical conditions. CSL's product areas include blood plasma derivatives, vaccines, antivenom, and cell culture reagents used in various medical and genetic research and manufacturing applications. The company was established in 1916 as Commonwealth Serum Laboratories and was wholly owned by the Australian federal government until its privatisation in 1994.

Antibody-dependent enhancement (ADE), sometimes less precisely called immune enhancement or disease enhancement, is a phenomenon in which binding of a virus to suboptimal antibodies enhances its entry into host cells, followed by its replication. The suboptimal antibodies can result from natural infection or from vaccination. ADE may cause enhanced respiratory disease, but is not limited to respiratory disease. It has been observed in HIV, RSV virus and Dengue virus and is monitored for in vaccine development.
GeoVax is a clinical-stage biotechnology company which develops vaccines. GeoVax’s development platform uses Modified Vaccinia Ankara (MVA) vector technology, with improvements to antigen design and manufacturing capabilities. GeoVax uses recombinant DNA or recombinant viruses to produce virus-like particles (VLPs) in the person being vaccinated.
A neutralizing antibody (NAb) is an antibody that defends a cell from a pathogen or infectious particle by neutralizing any effect it has biologically. Neutralization renders the particle no longer infectious or pathogenic. Neutralizing antibodies are part of the humoral response of the adaptive immune system against viruses, intracellular bacteria and microbial toxin. By binding specifically to surface structures (antigen) on an infectious particle, neutralizing antibodies prevent the particle from interacting with its host cells it might infect and destroy.
SAV001-H is the first candidate preventive HIV vaccine using a killed or "dead" version of the HIV-1 virus.
Dan Hung Barouch is an American physician, immunologist, and virologist. He is known for his work on the pathogenesis and immunology of viral infections and the development of vaccine strategies for global infectious diseases.
A molecular clamp is a polypeptide used to maintain the shape of proteins in some experimental vaccines. On a virus, pre-fusion proteins on their surface provide an attractive target for an immune reaction. However, if these proteins are removed or made by recombinant technology, they lose their shape and form what is called a "post-fusion form". When part of a virus, these proteins maintain their form by forming a quaternary structure with other viral proteins. The pre-fusion state of the protein is a higher energy metastable state. The extra energy is used to overcome the activation barrier of the fusion to the cell membrane. The virus protein in combination with the clamp polypeptide is called a chimeric polypeptide.

COVID-19 drug development is the research process to develop preventative therapeutic prescription drugs that would alleviate the severity of coronavirus disease 2019 (COVID-19). From early 2020 through 2021, several hundred drug companies, biotechnology firms, university research groups, and health organizations were developing therapeutic candidates for COVID-19 disease in various stages of preclinical or clinical research, with 419 potential COVID-19 drugs in clinical trials, as of April 2021.
Marylyn Martina Addo is a German infectiologist who is a Professor and the German Center for Infection Research (DZIF) Head of Infectious Disease at the University Medical Center Hamburg-Eppendorf. Addo has developed and tested vaccinations that protect people from Ebola virus disease and the MERS coronavirus EMC/2012. She is currently developing a viral vector based COVID-19 vaccine.
Jason S. McLellan is a structural biologist, professor in the Department of Molecular Biosciences and Robert A. Welch Chair in Chemistry at The University of Texas at Austin who specializes in understanding the structure and function of viral proteins, including those of coronaviruses. His research focuses on applying structural information to the rational design of vaccines and other therapies for viruses, including SARS-CoV-2, the novel coronavirus that causes COVID-19, and respiratory syncytial virus (RSV). McLellan and his team collaborated with researchers at the National Institute of Allergy and Infectious Diseases’ Vaccine Research Center to design a stabilized version of the SARS-CoV-2 spike protein, which biotechnology company Moderna used as the basis for the vaccine mRNA-1273, the first COVID-19 vaccine candidate to enter phase I clinical trials in the U.S. At least three other vaccines use this modified spike protein: those from Pfizer and BioNTech; Johnson & Johnson and Janssen Pharmaceuticals; and Novavax.

Medicago Inc. was a Canadian biotechnology company focused on the discovery, development, and commercialization of virus-like particles using plants as bioreactors to produce proteins, candidate vaccines, and medications. By using live plant leaves as hosts in the discovery and manufacturing process, the Medicago "Proficia" technology intended to create a rapid, high-yield system for its product candidates. Privately owned by a subsidiary of Mitsubishi Tanabe Pharma, Medicago and its product development programs were terminated by Mitsubishi in February 2023.

CoVLP was a COVID-19 vaccine developed by Medicago in Canada and GlaxoSmithKline (GSK). The product and Medicago, Inc. were owned by Mitsubishi who terminated the company and program in February 2023 due to high international market competition for COVID-19 vaccines.

The CureVac COVID-19 vaccine was a COVID-19 vaccine candidate developed by CureVac N.V. and the Coalition for Epidemic Preparedness Innovations (CEPI). The vaccine showed inadequate results in its Phase III trials with only 47% efficacy. In October 2021 CureVac abandoned further development and production plans for CVnCoV and refocused efforts on a cooperation with GlaxoSmithKline.

SCB-2019 is a protein subunit COVID-19 vaccine developed by Clover Biopharmaceuticals using an adjuvant from Dynavax technologies. Positive results of Phase I trials for the vaccine were published in The Lancet and the vaccine completed enrollment of 29,000 participants in Phase II/III trials in July 2021. In September 2021, SCB-2019 announced Phase III results showing 67% efficacy against all cases of COVID-19 and 79% efficacy against all cases of the Delta variant. Additionally, the vaccine was 84% effective against moderate cases and 100% effective against hospitalization.

A viral vector vaccine is a vaccine that uses a viral vector to deliver genetic material (DNA) that can be transcribed by the recipient's host cells as mRNA coding for a desired protein, or antigen, to elicit an immune response. As of April 2021, six viral vector vaccines, four COVID-19 vaccines and two Ebola vaccines, have been authorized for use in humans.

COVAX-19 is the result of a collaboration between Vaxine and CinnaGen, a private company with operations in the Middle East. COVAX-19 is a recombinant protein-based COVID-19 vaccine developed by South Australian-based biotech company Vaxine. It is under clinical trial in collaboration with the Iranian company CinnaGen.

John R. Mascola is an American physician-scientist, immunologist and infectious disease specialist. He was the director of the Vaccine Research Center (VRC), part of the National Institute of Allergy and Infectious Diseases (NIAID), National Institutes of Health (NIH). He also served as a principal advisor to Anthony Fauci, director of NIAID, on vaccines and biomedical research affairs. Mascola is the current Chief Scientific Officer for ModeX Therapeutics.









